 With the long-anticipated approval of Epidiolex, a proprietary form of cannibidiol (CBD) for two rare forms of childhood epilepsy, the Food and Drug Administration has put into question the future availability of CBD as a non-prescription dietary supplement.
With the long-anticipated approval of Epidiolex, a proprietary form of cannibidiol (CBD) for two rare forms of childhood epilepsy, the Food and Drug Administration has put into question the future availability of CBD as a non-prescription dietary supplement.
The landmark approval, announced on June 25, is the first time the agency has approved any product derived from cannabis, which the federal government still classifies as a Schedule 1 narcotic.
The move is a big win for Greenwich Biosciences, a subsidiary of GW Pharma, the English drug company that developed Epidiolex. It’s also a potential win for families of children with Lennox-Gastaut and Dravet syndromes—two severe forms of epilepsy for which there were no reliably effective drugs.
But it could throw a very wet blanket on the extremely hot market for non-prescription CBD supplements.
For years, FDA has made it clear that it considers CBD and other cannabinoids to be potential pharmaceuticals, not supplements. But by and large, the agency has not made a concerted effort to stop the sale of CBD supplements.
That could change following approval of Epidiolex. In a press conference immediately following the approval, FDA Commissioner Scott Gottlieb, MD, hinted at “a toolbox of enforcement actions” that the agency can take against companies marketing non-Rx CBD supplements.
Fast-Tracked Approval
GW convincingly proved that its proprietary CBD formulation could outperform placebo in a series of three randomized, double-blind trials involving more than 500 young patients with Lennox-Gastaut or Dravet’s syndrome.
FDA fast-tracked Epidiolex as an orphan drug for these very severe, albeit rare seizure disorders. Commissioner Gottlieb stressed that the cannabis drug development pathway is open to any company that identifies an unmet medical need, and is willing to invest in the requisite drug research and quality control.
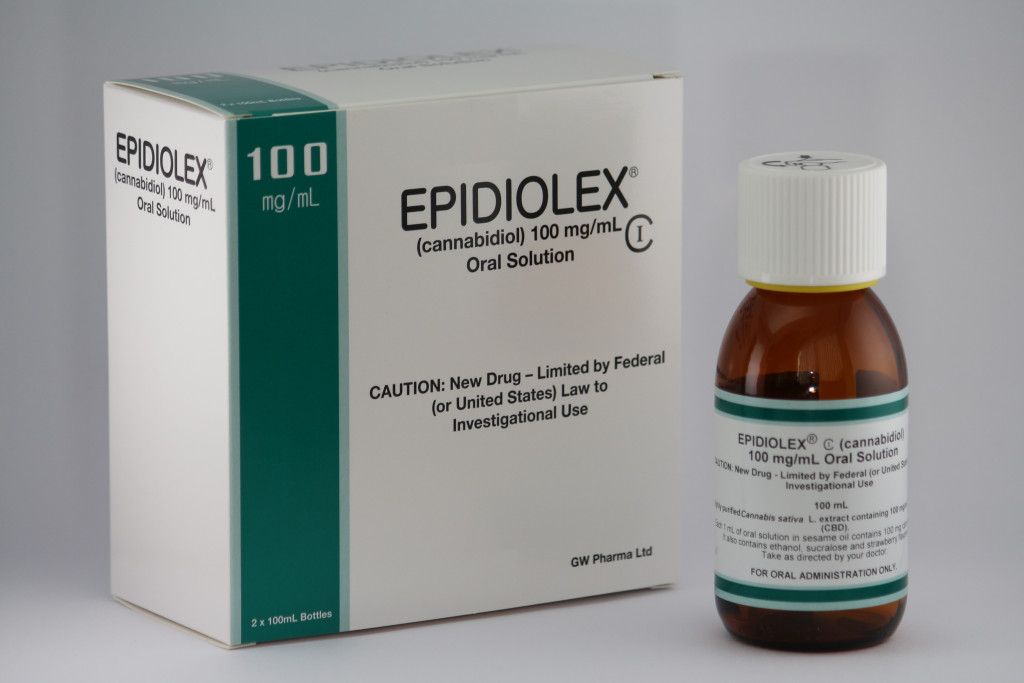 In an official statement, he said, “This product approval demonstrates that advancing sound scientific research to investigate ingredients derived from marijuana can lead to important therapies.”
In an official statement, he said, “This product approval demonstrates that advancing sound scientific research to investigate ingredients derived from marijuana can lead to important therapies.”
According to Gottlieb, the agency has had a strong interest in cannabis-derived drugs, and has taken specific steps to support cannabis research, including formation of a “Botanicals Team” to “provide expertise for researchers developing drugs derived from plants, such as marijuana.”
DEA to Reclassify CBD
It has also urged the Drug Enforcement Agency (DEA) to approve several hundred new licenses allowing academic and pharma industry researchers to legally study cannabis-derived substances.
“Drugs derived from marijuana also are eligible for several programs that are intended to facilitate and expedite development and review of new drugs that address unmet medical needs in  the treatment of serious or life-threatening conditions,” Gottlieb stated.
the treatment of serious or life-threatening conditions,” Gottlieb stated.
Now that there is an official medical use for CBD, the DEA will be obliged to delist CBD—or at least GW’s form of it—from Schedule 1 (drug of abuse with no legitimate medical benefit). Some cannabis advocates see this as a positive step toward eventual decriminalization of cannabis itself.
However, the way in which DEA will reclassify CBD remains to be determined. It is also unclear whether any reclassification will apply to CBD in general or only to branded and FDA-approved forms like Epidiolex.
FDA has made it very clear that approval of Epidiolex should not be construed as a broad FDA approval of marijuana or CBD: it is simply the approval of a proprietary formulation for a specific—and very narrow—clinical indication.
Freedom to Go Off-Label
Though Epidiolex was only approved for childhood epilepsy, physicians have some freedom to make off-label recommendations for its use to treat other conditions, according to FDA spokesman Michael Felberbaum.
“From the FDA’s perspective, with a few exceptions, health care professionals generally may choose to prescribe or use a legally marketed human drug for an unapproved use when they judge that the off-label use is medically appropriate for an individual patient,” Felberbaum told Holistic Primary Care.
“However, statutes and legal requirements administered by authorities other than the FDA also may govern prescribing, including, but not limited to, the Controlled Substances Act and state medical licensing and practice requirements.”
He stressed that FDA’s approval of a drug like Epidiolex for a particular disorder does not assure its safety and effectiveness for other uses. “A separate consideration of risks and benefits is necessary for each intended use to ensure that the expected benefits of using the product to treat a particular disease or condition are appropriate for its risks.”
Further, the regulatory status of non-branded, non-approved forms of CBD is unclear, so the protections that would apply for an off-label recommendation of Epidiolex may or may not apply to other non-Rx forms of CBD.
Can CBD be a Supplement?
The “pharmaceuticalization” of CBD will likely temper the billion-dollar market for CBD supplements.
This year’s Natural Products Expo West –a yearly gathering of over 80,000 people involved in the natural food and holistic health industries–made plain the extent to which CBD has  invigorated the dietary supplement market. Sales of CBD have soared in recent years. Retailers speaking at a CBD Summit preceding the Expo noted that CBD is moving off shelves as fast or even faster than less controversial supplements like omega-3s or probiotics.
invigorated the dietary supplement market. Sales of CBD have soared in recent years. Retailers speaking at a CBD Summit preceding the Expo noted that CBD is moving off shelves as fast or even faster than less controversial supplements like omega-3s or probiotics.
The surge is driven in part by consumers’ fears that once Epidiolex is approved, CBD will no longer be freely available without prescription.
The action is not only in the retail aisles. Several major practitioner-only supplement brands including Thorne Research and Designs for Health have already launched cannabis/hemp-based supplements containing CBD. Word is, several others are following suit.
Further, hemp companies like Tulsa-based Encore Life Sciences, have launched new practitioner-targeted CBD lines. And some wildly popular consumer-focused CBD brands like Charlotte’s Web are now exhibiting at integrative and functional medicine conferences.
The stellar growth has been despite the fact that the FDA has for years stated that it does not consider CBD to be a legal ingredient for dietary supplements.
Cannabis, Hemp, & Politics
At the federal level, cannabis is still illegal. Approval of Epidiolex does not change that fact.
Todd Harrison, an attorney with Venable LLP, who specializes in dietary supplement regulation, notes that according to the DEA, “marijuana,” means all parts of the Cannabis sativa plant— the seeds, the leaves, the stalks, the resin, everything.
 In the eyes of the law, cannabis and hemp are variations of the same plant, the sole difference between them being the relative content of tetrahydrocannabinol (THC). To be recognized as “industrial hemp” the plant—and any extracts derived from it—must contain no more than 0.3% THC.
In the eyes of the law, cannabis and hemp are variations of the same plant, the sole difference between them being the relative content of tetrahydrocannabinol (THC). To be recognized as “industrial hemp” the plant—and any extracts derived from it—must contain no more than 0.3% THC.
The Senate’s recent passage of the Hemp Farming Act as part of the Senate Farm Bill will exempt low-THC hemp from the federal definition of “marijuana” under the Controlled Substances Act, thus legalizing commercial hemp agriculture for a wide variety of uses. But this will not change the status of cannabis–the THC rich “drug-containing” variants.
The Farm Bill will redefine hemp as distinct from marijuana, and it will permit American farmers to grow as much hemp as they please. But it says nothing about the freedom to market hemp-derived products, “despite what the marijuana lawyers will tell you.”
The Hemp Farming Act was sponsored by Sen Mitch McConnell (R-KY), with an identical bill brought to the House. McConnell’s move induced euphoria among many hemp advocates, leading some to canonize the arch-conservative as a new patron saint of herb.
But as with nearly everything else today, hemp is a political football. McConnell represents an agriculture-heavy state where farmers need new opportunities. From that perspective, industrial hemp is a good bet. But the Kentucky senator has consistently affirmed his opposition to legalization of marijuana.
Sen. Chuck Grassley (R-IA), who is even more staunch in his disdain for cannabis, broke ranks with McConnell over the Hemp Farming Act, and introduced an amendment that would explicitly exclude “cannabinoids,” “extracts,” and “derivatives” from the Farm Bill’s definition of industrial hemp.
That basically means CBD.
Grassley’s amendments never made it to the floor, but the Iowa lawmaker says he remains committed to opposing any legislation that legalizes cannabis-derived substances.
In general, though, the Hemp Farming Act has bipartisan support—and it is one of the few issues these days that does. Senate minority leader Chuck Schumer (D-NY) is on board. And with the Senate’s passage, the House version of the bill will likley pass as well. Harrison, who works in DC, predicts that hemp legalization won’t meet opposition from the Executive Branch. “I don’t think Trump cares one way or the other.”
So, yes, hemp will likely be de-scheduled. But that probably won’t change things for CBD supplement makers.
That’s because CBD is derived from the resin of the cannabis plant.
And since, by DEA statutes, the resin counts as “marijuana” and not “hemp,” CBD counts as “marijuana” despite the wishes of the CBD industry. Low-THC hemp strains tend to produce very little CBD-containing resin. Which means that most CBD comes from straight-up cannabis, Harrison explained.
He added that any company claiming to have “seed-derived CBD oil” is selling hemp seed oil with added CBD.
A “Duty to Enforce”?
It might all sound like legal hair-splitting. But the words and definitions delineate the terms of engagement in the market, and the positions that the FDA and DEA are going to take.
GW had filed Investigational New Drug application for its CBD-based drug products years back in 2014. The company’s Sativex (Nabiximols)—an oral spray for use to treat pain and spasticity in people with multiple sclerosis—was approved as a drug in the UK in 2010.
The approval of Epidiolex concretizes the FDA’s position that since GW has developed CBD as a prescription drug, it cannot therefore be sold as a supplement unless a company can definitively prove that it was marketing CBD as a supplement or conventional food prior to GW’s IND’s.
That, says Harrison, is going to be a difficult feat.
The agency probably will not take issue with the sale of hemp seed or hemp seed oil as foods. These have been used as foods for decades, and contain very little CBD. But it will be much harder to make the case that hemp leaves or flowers—which do contain CBD-rich resin—are or ever were a food source for humans.
He expects that with the Epidiolex approval, FDA officials will feel a strong “duty to enforce” the hard line protecting prescription meds from unapproved non-prescription products.
Commissioner Gottlieb says the agency has already taken numerous actions against marketers making claims that CBD can treat cancer and other serious disorders. But these actions were based on the claims made, on the grounds that the Dietary Supplements Health Education Act (DSHEA) prohibits disease treatment or prevention claims for supplements of any kind.
It remains to be seen whether FDA will take action against CBD supplement companies on grounds that they are selling unapproved pharmaceuticals.
During the press briefing, Commissioner Gottlieb took the position that the pharma model ensures product purity and consistency of dosing, which increases the likelihood that patients will obtain the benefits observed in the clinical trials.
However, by categorically eliminating all CBD supplements, the FDA would put a formerly accessible non-prescription product out of reach of millions of people who currently use CBD  supplements for everything from chronic pain and PTSD to insomnia.
supplements for everything from chronic pain and PTSD to insomnia.
There are few reports of serious harm from self-directed use of CBD, though reporting of supplement-related adverse events tends to be very spotty.
If FDA chooses to take widespread action against non-Rx CBD (and has the budget to do so), anyone wishing to obtain it will need a prescription. Yet Epidiolex—the only existing CBD drug—was approved for a very narrow indication.
Impact on Practitioners
Are practitioners who dispense or recommend CBD supplements crossing legal lines?
They could be, says Harrison, though legal opinions on this matter are as varied as medical opinions about cannabis itself.
While regulators are generally unwilling to interfere with practitioner-patient relationships, or to challenge the “learned intermediary” status that medical professionals hold, they might feel more emboldened to do so now that there’s a newly approved Rx-only form of CBD.
Harrison believes physicians take some legal risk in “marketing an ingredient that the DEA considers a controlled substance.” He added that companies selling these products to and through doctors are also taking calculated risks.
Whether the laws are reasonable or not, whether they serve the best interests of practitioners or patients is certainly up for debate. What’s not debatable is the public’s interest in and desire for freely accessible non-Rx forms of CBD; the sales figures show this clearly.
From a practical point of view, what really matters is the scope and intensity of FDA and DEA enforcement of existing statutes around CBD and cannabis.
It remains to be seen whether the federal agencies will tolerate the re-naming of CBD as “whole plant hemp extract,” whether they will continue to permit widespread sale of non-Rx CBD, and whether they have the budgets and personnel needed to do anything more than issue a lot of strongly worded warning letters.
END







