 On July 30, former FDA Commissioner, Scott Gottlieb, published an op/ed in the Washington Post entitled, “The CBD craze is getting out of hand. The FDA needs to act.”
On July 30, former FDA Commissioner, Scott Gottlieb, published an op/ed in the Washington Post entitled, “The CBD craze is getting out of hand. The FDA needs to act.”
It’s a massive understatement given that there are now well over 1,000 companies selling supplements, foods, beverages, cosmetics, confections—even room diffusers—claiming to contain CBD. They promise myriad health benefits, most of which cannot be substantiated.
It’s also an ironic statement, coming as it does, from a man whose two-year tenure at the FDA will be remembered—rightly or wrongly—as the dawn of “legal” cannabis products.
Gottlieb presided over the approval of Epidiolex, the first cannabis-derived prescription drug approved for sale in the US. His time at the agency also overlapped Congress’ passage of the 2018 Farm Bill legalizing cultivation and processing of low-THC “industrial hemp” (as distinct from high-THC “marijuana”), and the Drug Enforcement Agency’s liberation of hemp from drug-of-abuse status.
Those moves pushed an already surging non-Rx CBD market into overdrive, with many new brands rushing in under a simplistic belief that the Fed had “legalized CBD.”
The truth is far more complex. The Farm Bill permits farmers to legally grow hemp with a THC content under 0.3%. But it does not formally legalize the sale of CBD or other hemp-derived substances (with the exception of hemp seeds and hemp seed oil) in foods, beverages, or supplements. Marijuana-derived CBD—extracted from plants containing greater than 0.3% THC—remains illegal.
The Farm Bill explicitly defers to the FDA on the matter of hemp-derived CBD.
FDA, for its part, has made clear that, for now anyway, it does not consider purified, isolated CBD to be a lawful dietary ingredient. The only “approved” and legal form of CBD is the highly-purified form patented by GW Pharmaceuticals as Epidiolex.
But the agency does leave a door open for CBD companies to apply for New Dietary Ingredient status, which would subject their extracts to thorough safety review. FDA is also encouraging companies to take the longer and costlier drug development route rather than opting for the supplement fast lane.
Hemp Extracts: A Grey Area
FDA’s existing rules also leave a grey area regarding hemp extracts that contain CBD along with other cannabinoids (but not THC), in their naturally-occurring, non-isolated, non-purified concentrations. Hence the proliferation of “full spectrum” or “whole plant” hemp oil extracts now flooding the market.
Many companies believe the “full spectrum” designation—a marketing term with no formal statutory definition—protects them from regulatory action. That may be more wishful thinking than reality.
In many ways, CBD epitomizes all the conflicting motives and scientific ambiguities with which the FDA must constantly contend: the pharma industry’s intention to protect its patents; the supplement industry’s wish to freely market “healthy” natural products; the public’s enthusiasm for non-Rx options; and the agency’s own mandate to protect the public from toxic, adulterated, or fraudulent products.
Absence of clear federal policy on CBD-containing hemp extracts has created tremendous confusion about what the government does—and does not—consider “legal.” This is further compounded by the fact that neither the Farm Bill nor the FDA’s rules override state level cannabis laws, which vary markedly.
The result? A patchwork of local and federal regulations and enforcement policies that baffle manufacturers, consumers, clinicians, and lawmakers alike.
Historic, Inconclusive Meeting
Last winter, the FDA announced a major initiative to clarify the rules for CBD.
On May 31, shortly after Commissioner Gottlieb’s unexpected resignation, the agency held the first in a series of public hearings to explore regulatory revisions that would simultaneously uphold the prescription sanctity of Epidiolex (and any future cannabis-derived drugs), while opening a channel for legal non-Rx hemp products.
Among the many issues on the table:
- Can CBD be a drug, a supplement, a food, and a cosmetic all at the same time?
- Should the commercial status (ie Rx vs non-Rx, drug vs supplement) of CBD be defined solely on the basis of concentration (pure CBD versus low CBD extracts)?
- How safe is CBD? For whom? At what dosage?
- What claims are permissible given the state of CBD science?
- Should synthetic CBD be permitted?
- Are available analytic methods adequate and fit-for-purpose to ensure safety and quality?
The meeting drew testimony from over 50 people—advocates and critics alike—representing the cannabis industry, supplement and food brands, consumers, medical practitioners, legislators, legal experts, and representatives of regulatory agencies.
The general consensus is that this historic hearing, though inconclusive, was a reasonable first step toward improving the current regulatory morass.
During a public comment period that ended on July 16, the agency received over 4,200 comments.
 Loren Israelsen, executive director of the United Natural Products Association, and one of the architects of the original DSHEA legislation, has reviewed the entire docket with his team.
Loren Israelsen, executive director of the United Natural Products Association, and one of the architects of the original DSHEA legislation, has reviewed the entire docket with his team.
“Roughly 10% of the comments we consider substantive. The vast majority are personal experience anecdotes from private citizens,” says Israelsen, adding that his organization like others are analyzing the comments in light of the FDA’s stated positions on CBD, as well as the broader context of the Farm Bill.
Patent Protection vs Public Demand
CBD is a booming market that will top $20 billion in US sales by mid-decade. And legalized hemp has opened up a vast new agricultural sector at a time when American farms are hurting. The food, supplement, beverage (alcoholic and non-alcoholic), tobacco, pet product, and mass market retail industries all have a stake in the FDA’s deliberations.
Nobody in Washington—including the FDA—wants to kill a Golden Goose of this magnitude.
But the FDA is also under obligation to protect the pharma industry and its investments. GW Pharmaceuticals, the maker of Epidiolex, is on record stating that in principle it has no objection to non-Rx low-dose CBD, provided that manufacturers adhere to strict quality standards, do not make disease claims, and do not borrow Epidiolex studies to support their products.
But many CBD marketers are stepping way over the line on all counts, and GW’s patience is wearing thin.
Alice Mead, GW’s VP of US Professional Relations, has indicated that if the food and supplement makers cannot agree to play within bounds on product claims and CBD levels, GW will become more assertive of its rights as an approved drug patent holder.
Israelsen says unapproved sale of purified CBD isolates as “supplements” is perceived as a threat to the entire drug approval system. It is likely to meet with significant pharma industry pushback in the coming months.
Mislabeling is Very Common
While some brands try to get away with selling purified pharma forms of CBD as supplements, others market products that contain little or no CBD.
In 2017, Marcel Bonn-Miller at the University of Pennsylvania and colleagues subjected 84 unique CBD products from 31 different companies—oil-based hemp extracts, alcohol tinctures, and vape liquids—to triplicate cannabinoid analysis.
They found a disturbingly wide range of CBD levels. On average, the products were labeled as containing 15 mg/mL of CBD, but actual product samples showed CBD levels ranging from 0.10 mg/mL to 655 mg/mL. The median was 9.5 mg/mL, far lower than the median label claim (Bonn-Miller MO, et al. JAMA. 2017).
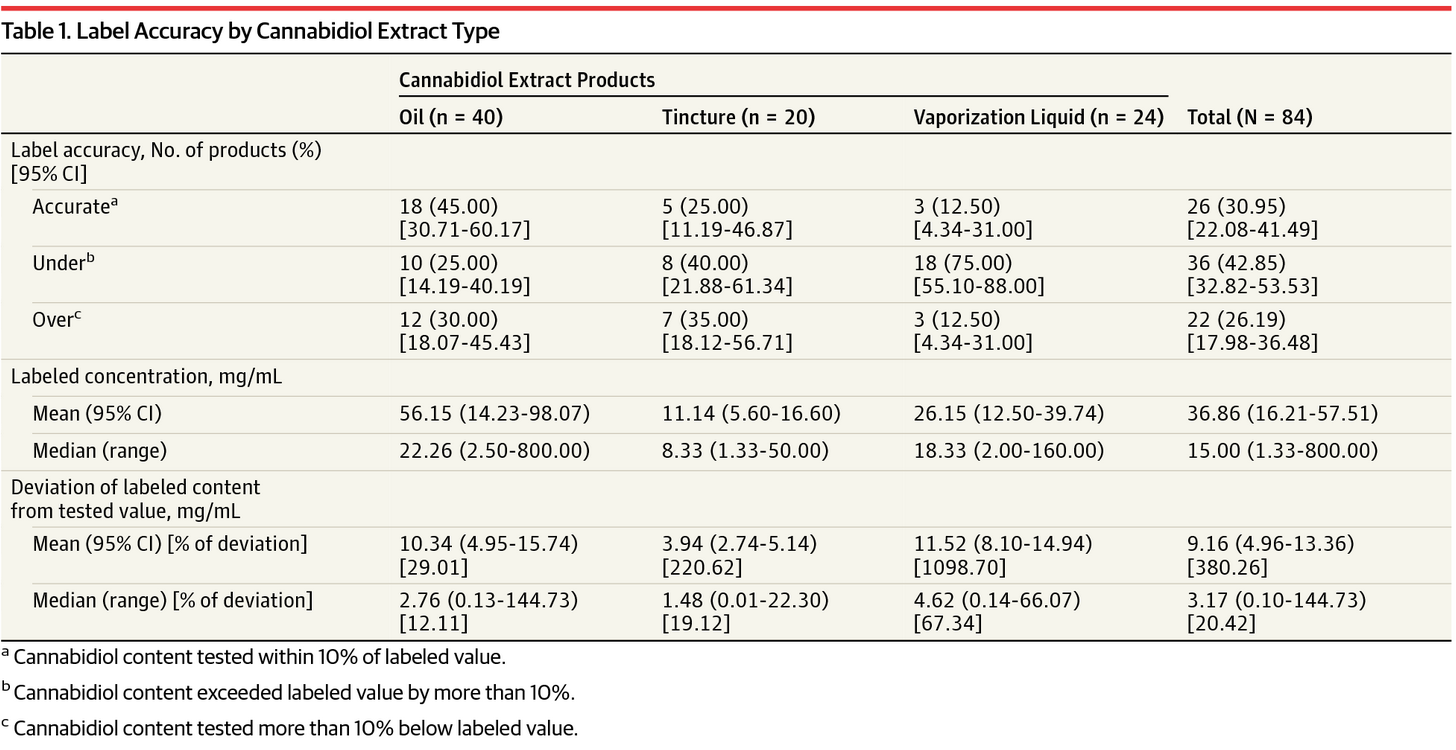
Twenty-two out of the 84 (26%) contained less CBD than indicated on the label, while 36 of the 84 (36%) exceeded their label claims. Only 31% were accurately labeled.
These findings echo a 2015 FDA analysis of 13 off-the-shelf CBD products that found only 2 were accurately labeled for CBD content.
An Herb Like Any Other?
“The May 31 meeting made it clear that the crack in the door is really for hemp extract, not CBD itself,” says Douglas “Duffy” MacKay, ND, Senior VP of Scientific and Regulatory Affairs for CV Sciences, one of the nation’s leading producers of CBD-containing hemp products.
“Hemp is a botanical, and DSHEA allows for botanical extracts in supplements.” The problem, says MacKay, is that FDA has not clearly defined an upper limit for CBD in supplements.
Epidiolex is 99% pure CBD, and contains no other cannabinoids. Many retail hemp extracts contain between 10% and 30% CBD along with other compounds that may or may not be disclosed. That difference explains: A) why the prescription drug is vastly more expensive than CBD supplements, and B) why Epidiolex studies cannot be cited to substantiate mixed-cannabinoid extracts with lower CBD levels.
“To some degree hemp is just another herb. I don’t mean to demean its importance in the marketplace, but it should be regulated like any other herb.”
—Michael McGuffin, president of the American Herbal Products Association (AHPA)
But that difference also begs the question of how much CBD puts a hemp extract into Epidiolex’s swim lane. At this point, nobody knows.
“The FDA has never done this for any other compound,” says MacKay, who was previously director of scientific affairs for the Council for Responsible Nutrition.
“They regulate by intended use and safety. Like with fish oil, it’s not the level of EPA or DHA that makes the product a drug or a supplement, it is about intended use,” he said, referring to Lovaza and Vascepa—two prescription fish oils approved for reducing triglycerides. It was not their omega-3 content that made them drugs; it was their disease treatment claims.
 Further, the 0.3% THC distinction that the Farm Bill makes between “hemp” and “marijuana” is arbitrary. MacKay believes the FDA is struggling to deal with a set of non-scientific definitions developed by Congress which, he pointed out, “is not a science-based organization.”
Further, the 0.3% THC distinction that the Farm Bill makes between “hemp” and “marijuana” is arbitrary. MacKay believes the FDA is struggling to deal with a set of non-scientific definitions developed by Congress which, he pointed out, “is not a science-based organization.”
Cindy Sovine, a hemp and cannabis lobbyist in the Colorado State Legislature, feels similarly.
“Marijuana is a slang term that just became a legal definition,” she said at New Hope Natural Media’s annual Hemp and CBD Summit last spring.
A former pharma industry lobbyist, Sovine stressed that hemp and marijuana are the same botanical genus, and differ only in the relative amounts of cannabinoids they produce. And there are well over 100 known cannabinoids.
“‘Industrial Hemp’ is really just a catch-all for everything else that’s not defined as marijuana,” she said, adding that each cannabinoid could potentially become a new therapeutic or health support product, once its physiological effects are better understood.
Complex Plant, Complex Issues
That prospect opens up another Pandora’s Box for the FDA. The agency’s regulatory system is well-designed for assessing and regulating isolated bioactive compounds, but it has difficulty with the inherent complexity of herbs. And cannabis/hemp is a complex plant.
But it’s complexity should not be held against it. “To some degree hemp is just another herb. I don’t mean to demean its importance in the marketplace, but it should be regulated like any other herb,” says Michael McGuffin, president of the American Herbal Products Association (AHPA).
A 40-year veteran in the herbal industry, McGuffin contends that the FDA needs to bring its authority to bear in enforcing basic good manufacturing practices (GMPs) and compliance with the safety regulations already in place.
“If you’re in the business of selling products that contain hemp, that contain CBD, you need to be completely aware of and understanding all the regulations that would apply to supplements made with echinacea, or chamomile, or turmeric, or any other herb. Those all apply to your facility today. And that’s really the starting point,” said McGuffin at the New Hope Hemp and CBD Summit.
AHPA has been at the forefront of the herbal quality assurance effort. It has generated numerous guidance documents, technical standards, and training events aimed at improving the safety, quality, and integrity of botanical products, including those derived from hemp.
McGuffin says one of the big challenges is that many current CBD brands are start-ups with no previous experience in producing herbal products. In many cases, they’re looking for a fast score on a hot commodity, and they are ignorant of the rules and standards to which established herbal companies adhere.
For their part, many legacy herbal brands have shied away from CBD because the regulatory status has been so ambiguous. But that will change. As the FDA clarifies the boundaries, “We’re going to start to see hemp product extensions from some of the long-established herbal companies, the ones retailers and practitioners already trust for making really high-quality echinacea, chamomile, turmeric.”
Beyond the obvious issues of product quality and prescription status, CBD and other cannabis-derived products present other challenges.
Banking and Merchant Services: Many banks and merchant service companies remain wary of hemp-related businesses, especially with regard to interstate transactions. This is especially true of medical and recreational marijuana businesses. Though the DEA has de-scheduled hemp, the shadow of “illicit drug” still hangs over it.
Advertising and Marketing: The FDA has made it clear that it does not authorize disease or symptom claims for non-Rx CBD. But it has not clarified what, if any, claims are acceptable. Since the agency doesn’t recognize CBD as a dietary supplement, the DSHEA allowance of structure/function claims may not apply. Or it may. Nobody’s sure. Since regulatory actions have been few and far between, many marketers are taking an anything goes attitude.
Clinical Research: Everyone is calling for more clinical research on hemp, cannabis, and their derivatives. But the DEA has been very slow to increase the supplies of marijuana for scientific study.
DEA still considers THC-containing marijuana a drug of abuse. Scientists wishing to study it—and interactions between THC and CBD are an important research topic—must go through a complex application process, and can only obtain study materials from the University of Mississippi, the sole authorized grower. The agency has repeatedly promised to approve more suppliers, but this has not yet happened.
Plaintiffs Attorneys: Wild Card Regulators
In whatever way the FDA ultimately decides to handle CBD, the reality is it will take years before any policy changes are enacted. This means confusion will reign for a long time.
And that, many industry watchers say, spells opportunity for class-action lawyers and ambitious attorneys general.
Justin Prochnow, an attorney with GreenbergTraurig, who specializes in FDA and FTC law, believes the CBD industry is extremely vulnerable. He expects a swell of FDA and FTC actions against companies making overt disease claims, as well as those stating their products are “phytocannabinoid-rich” or “CBD-rich.”
Those “rich” claims, according to the FTC, are reserved for foods or supplements that deliver at least 20% of the recommended daily intake (RDI) of a given nutrient. Since there are no RDIs for CBD or phytocannabinoids, these are technically false claims.
FDA and FTC warning letters are like chum for plaintiff’s attorneys. Prochnow and other industry experts expect to see a lot of them in the coming months, followed by a rapid wave of class action lawsuits.
Attorneys general are also sharpening their knives. In July, a coalition of 37 state AGs called on the FDA to “cooperate with the states in protecting consumers from false advertising on cannabis-derived products, such as CBD, and the potential hazards of consuming such products in certain populations.”
Even without revisions to the regulations, the FDA itself has been taking action—albeit only occasionally. It recently issued a warning letter to Curaleaf, a Massachusetts company marketing what the FDA considered unapproved CBD products with unsubstantiated cancer, Alzheimer’s disease, opioid withdrawal, and pain treatment claims.
The CBD phenomenon is one of the most complex and contentious natural products issues ever to confront the FDA, the FTC, and Congress. How the agencies resolve their regulatory policies for CBD will likely have great impact on the regulation of supplements, foods, and drugs for years to come.
END







