Decades before legislators in Colorado or California even began to consider medical marijuana, Israeli chemist Raphael Mechoulam was investigating the biochemistry and pharmacology of cannabis-derived compounds, and urging the global medical community to shed its preconceptions about this plant, and to take it seriously as a source of safe and effective medicines.
and pharmacology of cannabis-derived compounds, and urging the global medical community to shed its preconceptions about this plant, and to take it seriously as a source of safe and effective medicines.
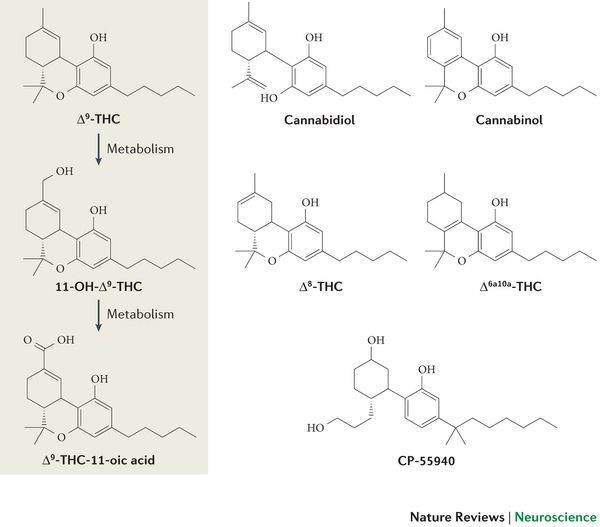
Mechoulam, now 86 and still active, is widely recognized as the grandfather of modern cannabis research. In the early 1960s, his team at the Hebrew University of Jerusalem became the first to isolate, characterize, and synthesize delta-9-tetrahydrocannabinol (THC), one of the main psychoactive compounds produced by cannabis plants.
His subsequent work included the discovery of endogenously produced cannabinoids and the characterization of the endocannabinoid receptor system within the human central nervous system and peripheral tissues.
Holistic Primary Care’s cannabis correspondent, Laura Lagano, had the privilege of interviewing Dr. Mechoulam during a recent US visit.
Lagano, an integrative clinical nutritionist and co-founder of the Holistic Cannabis Academy, got Mechoulam to share his views on the current state of medical cannabis research and the appropriate clinical applications of cannabis-derived compounds.
Here are exerpts from their far-ranging conversation:
Laura Lagano:Patients with medical marijuana recommendations—and many self-medicating adults without recommendations—seem to use cannabis for three main reasons: insomnia, mood disorders like social anxiety or depression, and pain. What do you think about those conditions in relationship to use of cannabis?
Raphael Mechoulam In Israel, cannabis has to be approved by the Ministry of Health, which is the like the FDA in this case. We have 26,000 patients that use cannabis for medical purposes. Most of them use it for pain, but not acute pain. If you’re going to have your tooth taken out, it’s not for that. But it’s for constant pain, neuropathic pain, pain that quite a few people have. And it helps. Most of the people, most out of these 26,000 patients, use it for pain, though not all of them, and yes, it definitely helps.
Anxiety is a very complicated situation and very difficult to measure. People use it. It helps, but then again, we need more data. The same is true for depression. We need better depression drugs. Depression in Patient A is not the same as depression in B, and C, and D, so we need a lot more clinical data.
LL: What do you think about marijuana or hemp-derived CBD (cannibidiol) for children with seizure disorders, neurobehavioral issues, cognitive impairment and the like?
RM: Many years ago, about 35 years ago, or even more, we did a clinical trial with cannabidiol. First, of course, we did a lot of work in mice. Then we found the  compound is not toxic, that the compound that can help with epileptic attacks in mice. So we went and did a clinical trial in human patients, (adults) not children. We had 15 patients. Half of them were uncontrolled. Half of them took cannabidiol, relatively high doses. And we were amazed how good it was.
compound is not toxic, that the compound that can help with epileptic attacks in mice. So we went and did a clinical trial in human patients, (adults) not children. We had 15 patients. Half of them were uncontrolled. Half of them took cannabidiol, relatively high doses. And we were amazed how good it was.
We found that out of the eight patients–eights patients that nothing was helping them anymore–out of these eight patients, four had no attacks while they were given cannabidiol. Three patients had much less, and only one was not affected. That was 35 years ago.
Now, I’m not a clinician. And at the university, we don’t have the ability or the money to do a big clinical trial. I expected that neurologists will pick up this initial work and expand it. Nothing happened. For 30-odd years, nothing happened. That’s a shame because we could have helped (a lot of) children or adults over that period of time.
I used to go to neurologists, who would say, “Well, we can’t do it. We don’t have the facilities. Cannabidiol comes from marijuana, from hashish. Of course, we will never get a permit.” So for 35 years, nothing happened until parents found that it helps their children, and they started giving them cannabis with high levels of cannabidiol, and in many cases, about 60, 70% of cases, it helped these children.
Now why did we have to wait for 35 years after an initial clinical trial? It was a small one, yes, but it showed that it works! Many drugs, many major drugs, started with a small trial.
It (clinical research) still doesn’t happen with most diseases where we know that cannabidiol might help. Yet we know that patients are using it without having a clinical trial. I don’t want to use a strong word, but it’s a shame that this has not happened.
LL: Well, I couldn’t agree with you more! As a parent of a child who has a seizure disorder and a developmental delay, I can tell you that when it’s your child, you want those results and you want that product, whatever it is, immediately. There’s no 35-year waiting period!
Do you hold any hope that US federal regulations will change, enabling US scientists and researchers to study this ancient herb and validate what you’re talking about and what we already know through testimonial?
RM: Well, I’m under the impression that things are changing. For many years, there were no clinical trials in the US, no major clinical trials. Now, things are changing, and I believe that there will be clinical trials, particularly with cannabidiol, less so with THC because THC has its side effects, but there are enough things to do at the moment with cannabidiol itself.
We had in Israel a clinical trial with a kind of autoimmune disease. In some cancer patients, the bone marrow has to be exchanged, and these people get bone marrow from the outside. The body doesn’t like it, and it attacks the bone marrow. It’s kind of an autoimmune disease, not real but a kind of autoimmune disease. The bone marrow that has been put in the body attacks the body. The body attacks the bone marrow, and the patient is sick, at times very sick.
from the outside. The body doesn’t like it, and it attacks the bone marrow. It’s kind of an autoimmune disease, not real but a kind of autoimmune disease. The bone marrow that has been put in the body attacks the body. The body attacks the bone marrow, and the patient is sick, at times very sick.
So a clinical group at one of the hospitals in Israel tried high doses of pure cannabidiol, very high doses, 300, 400 milligrams, and yes, it lowered many of the effects of this graft-versus-host disease. It most definitely works, and as a matter of fact, a company has taken it over and is developing it, so here we have something that has clinical trial background, and I hope that we shall see many more.
In another field, in schizophrenia, we showed many, many years ago in a few patients, that cannabidiol helps. A German group did quite a large clinical trial, very well done, published, and it showed that very high doses of cannabidiol, about 800 milligrams a day or even more, definitely helped with schizophrenia.
Now schizophrenia is a serious disease. About 1% of the population in the world has schizophrenia. Now that’s a lot, a lot of patients, and we need additional drugs. The drugs available now, they are pretty good. Some of them are excellent, but most cause side effects. Cannabidiol causes essentially no side effects, so we need more clinical trials in schizophrenia, but it has been shown that it works.
LL: An interesting fact in the US is that cannabis use is on the rise among the elderly, including people in long-term care facilities. What do you think is happening? Is there any potential to either prevent or reverse dementia? Is there a downside?
RM: It’s a complicated answer. We know that cannabis lowers anxiety. We know that cannabis lowers depression, mild depression at least. We have very little data on the effects of cannabis on Alzheimer’s, on dementia, and so on. There are papers, but not enough. It’s a very complicated situation.
We tried, and I was a little bit involved, giving cannabis to a group of (elderly) patients that were really in a bad shape. They were in a small community, about 20, 25 patients. They couldn’t eat by themselves. They couldn’t take care of themselves. They were in a bad shape. Most of them or many of them wouldn’t say anything. They would barely open their eyes. They wouldn’t say a word throughout a whole day and so on.
We started giving them small amounts of cannabis, which consisted of both THC and CBD, mostly THC. The amounts were not large. We gave them about five milligrams of THC, not pure THC, THC as it came with the cannabis (plant), two or three times a day. Quite a few of them seemed better. They opened their eyes. A few started saying a few words.
One of the patients who had been a writer previously said, “I feel fine,” and he started writing again. One of the patients, an elderly lady, said, “I feel fine,” and went home. So there is definitely an effect, but again, what kind of effect? What is their background? Do they have Alzheimer? Do they have something else? All these things have to be researched. We need the data. But yes, the answer is yes, it can help.
 LL: Do you see a place for both THC-containing cannabis and CBD-only cannabis in healthcare?
LL: Do you see a place for both THC-containing cannabis and CBD-only cannabis in healthcare?
RM: Both compounds are of extreme interest, but not for the same diseases. In some disease states, the THC is more important than CBD, and there are other disease states where CBD is more important. For example, in autoimmune diseases, in which the body attacks itself for various reasons, CBD seems to be much better than THC. For example, in diabetes type 1, in which the body attacks the cells that produce insulin, CBD is much more important than THC. In other disease states like trauma, apparently, THC is more important.
LL: What do you think about cannabis for up-regulating the endocannabinoid system for health and wellness, not just for treating diseases?
RM: The endocannabinoid system is of extreme importance. On the health side, there was just a review recently by senior people at NIH. They wrote that the endocannabinoid system is involved in essentially all human diseases. So obviously, it’s of extreme importance. Does it help situations that are not really clinical? Most of us are anxious at some point or another, or slightly depressed at one point or another. These are not clinical situations. These are situations of every day, and chances are that, yes, cannabidiol can affect that. CBD most definitely affects anxiety, mostly definitely helps with depression. So if it’s extreme, then of course, it’s a disease, but many of these things are not extreme, and in this case, CBD I believe can help.
LL: In the past, you’ve been critical of legalization for recreational use. Have your views changed?
RM: Well, I’ve not been critical. I just state that the medical use and the recreational use should be separate. The medical use should be based on medical data, and it should be advanced as modern drugs are advanced: through animal trials, through clinical trials, through everything that a new drug needs.
The recreational use is a social issue. If the population of a certain state, certain country, wants to have cannabis open to everybody or to most people after a certain age, then that’s fine. But it’s a social issue. Our population decided centuries ago that tobacco is fine. Of course, now we know that tobacco is a toxic agent. But it’s a social issue. Society has decided it’s okay. You can use it if you wish. The same is true for (recreational) cannabis.
LL: What’s your opinion about cannabis as an exit drug from addiction, particularly as it pertains to opioid addiction?
RM: People say cannabinoids or cannabidiol are good in order to lower opioid addiction. Now, addiction is a complicated state, whether we are speaking of the withdrawal symptoms, whether we are speaking of the actual addiction process, and so on. There are no clinical trials. So there are individual cases, and people say, “It has helped me with my addiction problem.” But we don’t have good clinical basis, so yes, I believe that cannabidiol is helpful, but it’s a yes with a question mark.
LL: We have a lot of athletes, particularly football players, promoting the use of cannabis as an exit from their opioid addiction, which was caused by opioids prescribed when they were players, to get them back on the field. I know many of these men say the same thing: “Cannabis saved my life.” Obviously, a clinical study this does not make, but oftentimes testimonials are the way we get to clinical studies. So hopefully, we’ll be seeing that in the future.
RM: Well, American football has a lot of problems, and some of the problems are head injury, closed head injury. Some of the people that smoke cannabis after football take it for the head injury rather than as an anti-opioid compound. CBD, in mice at least, is a good agent against the head injury. We’ve tried that. I collaborated with a professor who’s a specialist in this field, and she has found that, yes, CBD definitely helps with brain injury. Quite a few other compounds do the same, but again, we don’t have a clinical trial.
LL: Lack of cannabis education among both health professionals and consumers is certainly a challenge. Nobody in the US learned about the endocannabinoid system in medical school. I certainly didn’t learn about it in nutrition school. Do you have any thoughts about how this problem should be approached?
RM: It (endocannabinoid system) is going to be there (in medical education) within not decades but just a few years because it’s a major, major system. People speak of the major physiological systems. They have to speak of this one. The endocannabinoid receptors in the brain are present in larger amounts than any other receptor.
We do not know enough about them, but we know that endocannabinoids are active. There is a lot of work in omega-3 fatty acids. Well, the derivatives of omega-3 fatty acids are closely related to the endogenous cannabinoids. So these things will be tied together. I believe that within the next few years, these will be major topics in medical schools, in nutrition and so on. We just need to know more, and again I go back, we need to have also the clinical trials that will help.
To learn more about the Holistic Cannabis Academy’s training programs for physicians and allied health professionals who want to understand the role of the endocannabinoid system in health, and the practical applications of cannabis in clinical practice, visit: http://holisticcannabisacademy.com/?oprid=6013
END







