Co-enzyme Q10 is an essential, fat-soluble benzoquinone synthesized in mitochondrial membranes and found in nearly every cell in the body. Its prevalence in just about every tissue is what gave the substance its name: Ubiquinone–the ‘ubiquitous Quinone’.
body. Its prevalence in just about every tissue is what gave the substance its name: Ubiquinone–the ‘ubiquitous Quinone’.
Because it plays a vital role in mitochondrial electron transport, it is a linchpin in cellular energy production. The molecule was discovered in 1957, and 15 years later, CoQ10 deficiency was linked to congestive heart failure (CHF). The severity of CHF correlates strongly with the severity of CoQ10 deficiency, a finding that roused interest among cardiologists—and many people with heart disease.
CoQ10 is not just ubiquitous in the body; it is found on retail shelves in just about every place that sells dietary supplements.
Over the years, it has become one of the most popular non-vitamin supplement ingredients, taken by millions of people hoping to reduce cardiovascular risk, increase overall energy levels, combat fatigue and in some cases, mitigate statin-induced myopathy.
The research on CoQ10 has been highly variable. There’s no consensus on whether it truly prevents or mitigates heart disease. A 2014 Cochrane Collaboration metanalysis of 7 small CHF studies involving more than 900 patients, states that, “No conclusion can be drawn on the benefits or harms of coenzyme Q10 in heart failure at this time.” The limited size of the studies, inconsistencies between endpoints measured, heterogeneous patient cohorts, and inherent biases made it difficult to draw firm conclusions (Madmani ME, et al. Cochrane Database of Systematic Reviews, 2014).
That same year, data from Q-SYMBIO—a 2-year, multicenter study of 420 patients with moderate to severe CHF —showed that adding CoQ10, 100 mg thrice daily, to conventional drug therapy reduced CV mortality by 42%, and all-cause mortality by 44%. It also reduced CHF-related hospitalizations (Mortensen, et al. JACC Heart Failure. 2014; 26).
Many clinicians are not waiting around for a definitive nod from academic researchers. They see little harm and much potential benefit in recommending CoQ10 to patients with existing CVD or high-risk profiles.
“I’ve used CoQ10 since 1982. I did research on it in the 90’s, I’ve written books on it, I’ve published many peer-reviewed papers on it. And I can tell you this: after practicing cardiology for over 40 years, it would be unthinkable for me not to give CoQ10 to my cardiac patients. I feel it’s that valuable,” says Stephen Sinatra, an integrative cardiologist who directs the New England Heart Center in Manchester, CT.
The key, Sinatra says, is in getting the circulating blood levels up to at least 2.5 ug/ml. It is imperative to measure Q10 levels before supplementation, and 3 or 4 weeks after, and equally important to use a highly bioavailable form of the compound.
“Once you know you have an adequate blood level, then CoQ10 does its magic,” says Sinatra. At levels above the 2.5 ug/ml threshold, it confers therapeutic benefits for almost all patients, even those with advanced heart disease. At levels below 2.5, one will not likely see much impact. This, he says, is one reason CoQ10 studies have been so divergent or inconclusive. In many of the trials, researchers did not monitor or report blood levels.
The oral dosage needed to drive blood levels up over 2.5 will vary from patient to patient. In general, healthy people need less, and sicker people need more. Dr. Sinatra says it can range anywhere from 30-40 mg, thrice daily, for a healthy patient with cardiac arrhythmia or hypertension, to 300-500 mg daily for a patient with advanced CHF. He is not afraid to recommend up to 1,000 mg/day or more if repeated tests show inadequate CoQ10 blood levels at lower doses.
Ubiquinone vs Ubiquinol
The subject of CoQ10 bioavailability is a cause of contention and confusion for clinicians and patients alike. In its solid form CoQ10 is not highly absorbable. Consequently, formulators have developed a wide range of biochemical fixes—oils, gels, nanoparticles—to improve uptake. The market is rife with claims and counter-claims about optimal bioavailability.
One of the biggest debates centers around which basic form of CoQ10 is most effective: Ubiquinone or Ubiquinol.
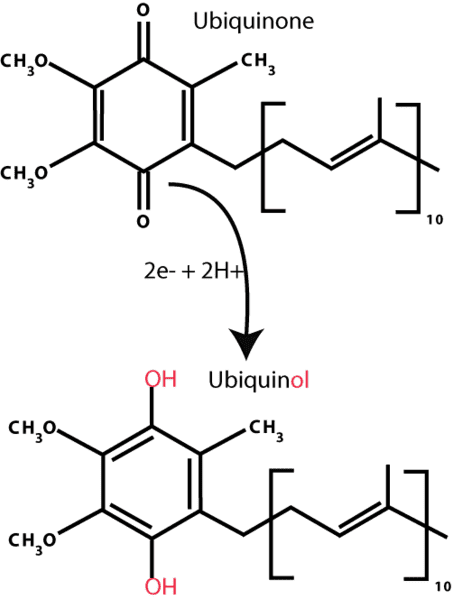 CoQ10 is a molecule that can readily switch between its oxidized (Ubiquinone) and reduced (Ubiquinol) forms. The difference is in the presence or absence of two electrons. The terms “ubiquinone” and “CoQ10” are sometimes used interchangeably, in part because prior to 2006, Ubiquinone was the only form available in supplements.
CoQ10 is a molecule that can readily switch between its oxidized (Ubiquinone) and reduced (Ubiquinol) forms. The difference is in the presence or absence of two electrons. The terms “ubiquinone” and “CoQ10” are sometimes used interchangeably, in part because prior to 2006, Ubiquinone was the only form available in supplements.
Technically—and physiologically—Ubiquinol and Ubiquinone are different. Ubiquinol is generally considered to be the “active” form. When it comes to choosing supplements, the big question is whether the differences matter clinically.
One could say that Ubiquinone is the “tried-and-true” form—the one with which most people are familiar. It is also the most studied form; all CoQ10 research prior to 2006 was of necessity done with Ubiquinone.
That changed when Kaneka—the world’s largest Q10 ingredient supplier, and the company that initially introduced Q10 to the supplement market—developed technology for commercial production and encapsulation of the reduced, electron-rich form.
Ubiquinol’s two additional electrons make it one of the strongest fat-soluble antioxidants. Kaneka promotes the ingredient as significantly more bioavailable than ubiquinone, a message amplified by supplement makers that utilize Kaneka’s raw materials. They suggest that Ubiquinol is the preferred choice for older, sicker people who have an impaired ability to synthesize CoQ10 and to convert the oxidized (Ubiqiunone) to the reduced (Ubiquinol) form.
The Age Factor
According to Robert Barry, PhD, Kaneka’s director of scientific affairs, CoQ10 levels naturally tend to drop as people age. “Some reports state that this decline in CoQ10 becomes apparent around 40 years of age, while other reports state it can occur as early as 20 years, with slow but continuous decline.”
He added that the ability to absorb CoQ10 from dietary sources also dwindles as we age, and the Ubiquinol to Ubiquinone ratio tends to shift as well. Over time, the ability to covert the oxidized to the reduced forms also declines. This indicates a lower antioxidant capacity and greater susceptibility to oxidative stress.
Dr. Barry holds that Ubiquinol is the best form of Q10 for anyone over 30 years old.
The claim is debatable.
Many things can diminish blood levels of CoQ10. Statins are an obvious culprit. Taken by well over 30 million people—these drugs reduce cholesterol by blocking the same hepatic pathways involved in synthesis of CoQ10. This effect can decrease the total CoQ10 levels by up to 40%. Diseases associated with oxidative stress like COPD, Parkinsonism, coronary artery disease, and some forms of cancer, can also deplete CoQ10 or cause shifts in CoQ10 redox status.
Last year, a team of German researchers published a study of 860 men and women, showing an inverse U-shaped relationship between CoQ10 concentration and age.” In general, women have lower levels than men at all ages, and older people tended to have lower levels than younger people (Niklowitz P, et al. J Clin Biochem Nutr. 2016; 58(3): 240-245). But not all the research on age and Q10 levels shows a decline: some studies indicate that total plasma CoQ10 levels stay relatively stable over the decades. There’s some data to suggest that levels may actually increase with age.
That said, most studies point to an increase in the ratio of Ubiquinone to Ubiquinol (the redox state) as we age (Miles MV, et al, Clinica Chimica Acta, 2004, Wada, J Am Geriatr Soc. 2007).
The relatively higher percentage of ubiquinone seen in older adults may be because of the body’s diminished ability to reduce the oxidized molecule due to age, as Kaneka claims. But just as likely, it could indicate increased oxidative stress and higher need for antioxidant protection.
Kaneka states that its Ubiquinol has significantly higher absorption and bioavailability than Ubiquinone, making it the best choice for people with chronic inflammation or an inflamed digestive tract. They support the bioavailability claim primarily with a study of male rats given comparable doses of Ubiquinol (95% reduced, 5% oxidized) and Ubiquinone (100% oxidized).
By the second hour, the animals given Ubiquinol had markedly double the amount of CoQ10 in their blood–4 ug/ml versus 2 ug/ml. From the sixth hour onward, CoQ10 levels were nearly identical between both groups. (Tatsumasa Mae, U.S. Patent No. US 6,184,255 B1, 2001)
It is important to note, however, that the Ubiquinol supplement used was 95% Ubiquinol, which is more concentrated than the 20-80% Ubiquinol found in most commercial supplements.
The sharp initial spike does suggest that Ubiquinol has a bioavailability advantage, at least at first. But the researchers do not address the leveling-off observed several hours later.
Clinically, there are examples of patients not responding well to Ubiquinone, and finding better results when switching to Ubiquinol. Peter Langsjoen and colleagues published a report summarizing 7 case studies of patients with stage IV CHF. In all cases, patients who had not had any measurable benefit from Ubiquinone showed significant changes—including ejection fraction increases of 10%-60% –shortly after they began taking Ubiquinol.
These patients had been supplementing with various doses of ubiquinone along with conventional drugs for 10 years, and had not been able to reach therapeutic blood levels of CoQ10 (Langsjoen PH, Langsjoen AM., Biofactors, 2008, p. 119-127).
In this particular study, all seven patients were able to reach therapeutic blood levels within months of switching to Ubiquinol. The average ejection fraction went from 22% to 39%, and their average NYHA CHF class dropped from IV to II.
The findings are impressive, but the study is very small and uncontrolled, and the authors do not disclose the type(s) of Ubiquinone the patients took prior to making the switch. So it is difficult to draw a firm conclusion from Langsjoen’s work.
Ubiquinol is more expensive than Ubiquinone. Dr. Sinatra says he’s yet to be convinced that the former is truly superior, and worth the added cost.
In a post on his website, he states: “The biggest claim with heart vitamins is that ubiquinol CoQ10 can be absorbed up to eight times better than other forms. Yet, I haven’t seen solid evidence to back up that claim. In fact, for years I’ve used the ubiquinone form with great success.”
Assuming an individual can efficiently convert the oxidized to the reduced form, both ubiquinol and ubiquinone should provide equivalent antioxidant and metabolic effects.
In specific cases, when a patient simply cannot obtain high serum levels after several months on relatively high doses of Ubiquinone, it would make sense to consider a switch. But make sure to do a thorough workup to exclude other reasons for the chronically low level, such as systemic inflammation or tissue damage.
Researchers and practitioners may not agree on everything about CoQ10, but here are a few basic points that are supportable by the available science:
-
CoQ10 supplementation can increase survival and reduce morbidity in CHF patients, if plasma levels reach the range of 2.5 – 3.5 ug/ml or greater. Lower levels tend not to be effective.
-
Supplementing with smaller, more frequent doses tends to improve bioavailability: 100 mg twice daily will yield higher results than 200 mg daily.
-
The higher the CoQ10 dose taken, the lower the percentage absorbed.
-
CoQ10 is not well absorbed in solid form. This includes tablets, granules, capsules, and suspensions. If using a solid form, a considerably larger amount of CoQ10 than actually needed must be administered to the patient.
-
There are no significant drug interactions or adverse reactions associated with CoQ10 supplementation. Generous dosing is generally safe, if needed to achieve therapeutic results.
END







