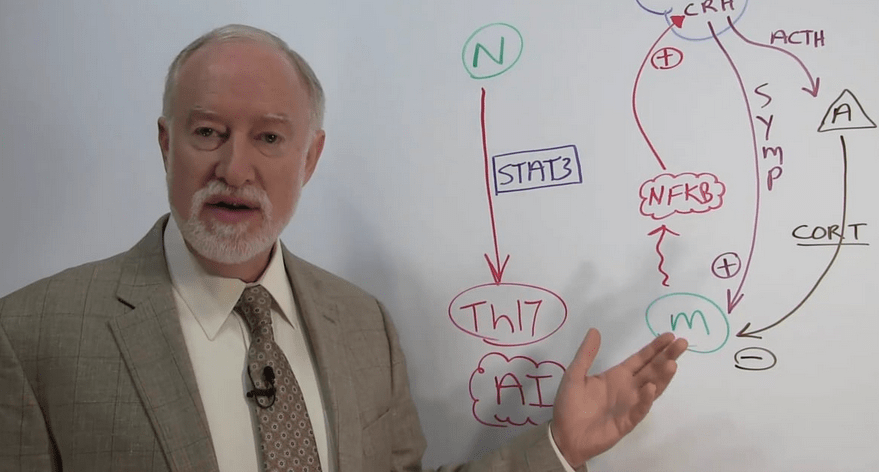
 When treating patients with complex autoimmune diseases, it’s advantageous to “think of the patient’s biology like a landscape,” proposed Samuel Yanuck, DC, FACFN, at the Institute for Functional Medicine’s 2018 Annual International Conference in Hollywood, FL.
When treating patients with complex autoimmune diseases, it’s advantageous to “think of the patient’s biology like a landscape,” proposed Samuel Yanuck, DC, FACFN, at the Institute for Functional Medicine’s 2018 Annual International Conference in Hollywood, FL.
Imagine a meadow. An “autoimmune flare is a ditch in the meadow,” he suggested.
As you observe your autoimmune patients walking through the “fields” of their lives, the question to ask is: “How far are they from the ditch?” You want to do your best to help these patients stay as far away from the ditch as possible.
Yanuck, director of the Yanuck Center for Life & Health in Chapel Hill, NC and adjunct assistant professor at the University of North Carolina School of Medicine, uses the landscape analogy to conceptualize autoimmune patients’ likelihood of experiencing disease flares. If a patient who is relatively far from the ditch experiences a perturbing event — like eating a trigger food, for instance, or contracting an infection — the incident might move her closer to the edge, but she won’t fall in. She won’t get an autoimmune flare.
But if that same patient–owing to ongoing stress, persistent unhealthy lifestyle choices, or toxic environment–is already near the ditch when a triggering event occurs, “then the very same increment of change is going to give them a flare — they’re going to fall in.”
Yanuck encouraged clinicians to teach patients how to identify and avoid the holes in their unique biological terrain. In doing so, “your ability to keep patients out of their autoimmune flare process will be greatly enhanced.”
Autoimmunity Begets Autoimmunity
What causes a patient to land in the ditch? A host of triggers, some related to specific autoimmune conditions and others more general, can play a role. The key point is that autoimmunity begets autoimmunity.
Heightened inflammation is a hallmark of autoimmune disease. It amplifies existing autoimmune processes, and can trigger development of new autoimmune disease. Therefore lowering inflammation is essential. It is equally important that patients already diagnosed with an autoimmune condition recognize the increased risk of developing a second. “They can get another one, and they need to know it, because they need to be motivated” to adhere strictly to treatment plans.
Inflammasomes: Inflammatory Suicide
One of the most important things to emerge from recent immunological research is the concept of the inflammasome.
Inflammasomes are pinwheel-like intracellular protein complexes that detect and respond to a range of inflammation-inducing stimuli, including microbial, stress, and tissue damage signals. Until they are called to action,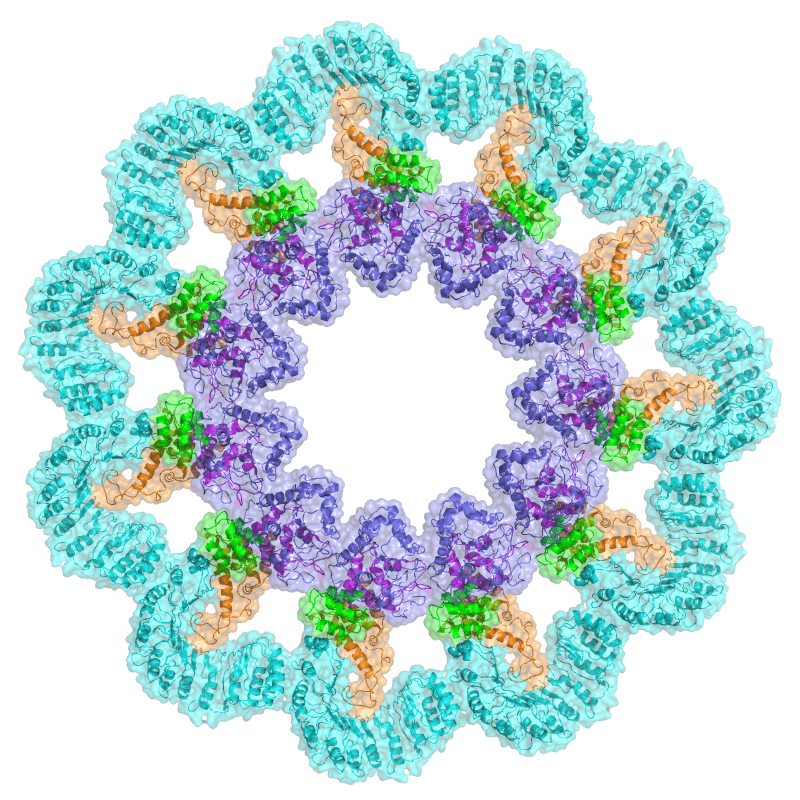 these multi-protein aggregations are disassembled as tiny structures inside cells. When a cell receives threat signals–such as from a pathogenic bacterium–it reassembles the inflammasomes, which in turn activate the releae of pro-inflammatory cytokines like interleukin-1β (IL-1β) and IL-18.
these multi-protein aggregations are disassembled as tiny structures inside cells. When a cell receives threat signals–such as from a pathogenic bacterium–it reassembles the inflammasomes, which in turn activate the releae of pro-inflammatory cytokines like interleukin-1β (IL-1β) and IL-18.
Inflammasomes also induce a form of programmed cell death known as pyroptosis. In this process, immune cells such as macrophages or T-helper cells respond to the presence of pathogens by releasing large amounts of inflammatory cytokines, and then swell themselves up until they burst en masse. In circumstances of acute infection, pyroptosis plays an important role in helping to eliminate pathogenic organisms by eliminating their replication niches and amplifying the host immune response.
In healthy patients, inflammasome activity regulates inflammation during microbial infections and plays vital roles in mucosal immune responses. But inflammasome overactivation has “dire consequences,” write the authors of a 2012 Nature review paper. It leads to widespread cellular destruction, diminishing the body’s innate Th1 immune response and natural killer cell counts. The authors link inflammasome dysregulation to the pathogenesis of obesity, atherosclerosis, inflammatory bowel disease, metabolic syndrome, and pancreatic beta cell death (Strowig, T. et al. Nature. 2012; 481(19): 278-286).
But like all defense mechanisms, pyroptosis can easily go awry and become chronic, thus contributing to systemic inflammation and autoimmunity.
“When you have inflammasome activation, you have cells essentially dying by an inflammatory suicide mechanism…they go up in flames and trigger their neighbors to go up in flames,” Yanuck described. Curbing the destruction mediated by active inflammasomes “will help with a big increment of movement away from the ditch,” Yanuck said.
Autoimmunity or Autoinflammation?
“We all talk about autoimmunity — but there’s a whole class of autoinflammatory diseases” that receive much less attention, he noted.
Rather than separating them into two distinct categories, Yanuck described a spectrum of related conditions, with autoimmune diseases affecting the adaptive immune system on one end, and autoinflammatory diseases marked by exuberance of the innate immune system on the other.
The “classic” autoinflammatory conditions like Mediterranean fever and other periodic fever syndromes are rare, and physicians tend to think of autoimmune and autoinflammatory disease as discrete conditions. But Yanuck pointed out that “every autoimmune patient has some percentage component of an autoinflammatory process, and attending to that gives you a big chunk of movement away from the ditch.”
It makes good sense to assess inflammasome activation in any patient with an autoimmune condition, and it is fairly easy to do.
A couple of clinical clues can tip you off to inflammasome overactivity: One is a surge in C-reactive protein (CRP), and the other is a low glomerular filtration rate (GFR).
“When you see a super high CRP with no obvious cause, you really want to think about autoinflammatory activation, inflammasome activation,” Yanuck told the IFM audience.
 Similarly, a low glomerular filtration rate (GFR) can also indicate inflammasome dysregulation. Low GFR reflects poor kidney function, which, in the absence of clear kidney disease, should be taken as a warning signal. All too many patients with autoimmune conditions like rheumatoid arthritis end up later in life on kidney dialysis. Yanuck cautioned that patients middling with their management of autoimmune conditions like rheumatoid arthritis “may have trouble later in life in a way with nothing that has to do with their autoimmunity.
Similarly, a low glomerular filtration rate (GFR) can also indicate inflammasome dysregulation. Low GFR reflects poor kidney function, which, in the absence of clear kidney disease, should be taken as a warning signal. All too many patients with autoimmune conditions like rheumatoid arthritis end up later in life on kidney dialysis. Yanuck cautioned that patients middling with their management of autoimmune conditions like rheumatoid arthritis “may have trouble later in life in a way with nothing that has to do with their autoimmunity.
“You have to deal with these inflammasomes.”
The key is to help patients build a well-supported innate immune response that is “highly efficient and not being negatively affected by excessive inflammatory signaling.”
To attenuate inflammasome activity, he recommended a number of natural treatments including: Resveratrol, sulforaphane, quercetin, curcumin, and potassium. All aid with various aspects of immune regulation, from halting inflammasome assembly to inhibiting NF-kB.
NF-kB & Neutrophils
Nuclear Factor (NF)-κB is a family of transcription factors involved in the immune response. It has long been a target in the treatment of inflammatory disease. Among its other functions, NF-κB induces the expression of various genes that encode highly inflammatory cytokines like IL‐1β and tumor necrosis factor alpha (TNFα).
For this reason, many clinicians paint NF-κB as the primary factor influencing inflammation. Research over the last decade is shifting that view. Dr. Yanuck stresses that the true problem is not so much the activation of NF-κB pathways — it’s the increased IL‐1β and TNFα production and the resulting influx of neutrophils that are all prompted by NF-κB.
He believes neutrophils should be a focal point for treatment of autoimmune disease.
Neutrophils are the most abundant white blood cell type in the body. In the presence of microbial pathogens, they initiate the immune response. Later, large macrophages consume the neutrophils and clear them from the affected tissue.
In patients experiencing chronic inflammation, this process is disrupted.
If too many neutrophils enter the tissue but are not cleared quickly enough, they die and burst open, releasing their contents — chiefly adenosine triphosphate (ATP) — into the surrounding tissue. ATP becomes a highly inflammatory substance when it leaves the cell. “When a cell senses that ATP is coming to its receptors, it knows that its buddies are dying. And when its buddies are dying, that’s a terrible signal, like something’s really wrong here,” Yanuck explained.
Dying cells send a message to the immune system that ultimately draws even more neutrophils to the affected area. When these new neutrophils arrive on the scene but aren’t cleared by the macrophages, they themselves cause additional damage to the extracellular matrix and surrounding tissue. That, in turn, signals for migration of even more neutrophils.
This is central to the ongoing cycle of chronic inflammation, and “resolving this is very much core to the whole endeavor” of autoimmune disease treatment, Yanuck says.
To curb inflammation, one needs to interrupt the primary signals that attract the neutrophils: that means IL‐1β and TNFα. Yanuck contends that the majority of chronic inflammation clinicians see in day-topday practice is the type fueled by neutrophils and triggered by IL‐1β and TNFα.
Notably, IL‐1β also shares an important connection with inflammasomes. “When the inflammasome assembles, before the cell dies, it pumps out a huge amount of IL‐1β,” Yanuck said. “That causes neutrophil recruitment into the region.”
Know Your Neutrophil Count
The first step in down-regulating this inflammatory cycle is to assess patients’ neutrophil counts. “Every time you pull blood out of a vein, you should do a CBC,” he urged. “It’s going to tell you a huge amount.”
In the absence of an obvious origin like an infection, a high neutrophil level — nearing or exceeding 70%, for instance — on a complete blood count indicates inappropriate immune system activation. With an infection, a short-lasting elevation in neutrophils is normal and expected. But physicians must recognize when this is indicative of an autoimmune flare. A climbing neutrophil count in a non-infected, non-injured person is a sign of ongoing chronic inflammation, and it should be a major red flag to clinicians.
One tool Yanuck recommends for attenuating neutrophil influx is fish oil. “Giving fish oil is a fantastic thing to do,” he said. It helps move patients “from the inflammatory process to the resolution phase.”
We commonly hear about the anti-inflammatory benefits of eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), the major essential omega-3 fatty acids abundant in fish oil — but what makes them so beneficial? A 2010 Journal of Internal Medicine paper authored by researchers at Brigham and Women’s Hospital and Harvard Medical School offers an explanation.
The authors write that omega-3 fatty acids, in particular EPA and DHA, are “precursors to a new genus of potent lipid mediators (LMs) that are both pro-resolving and anti-inflammatory…and serve a physiological role defining programmed resolution.” (Norling LV & Serhan CN. J Intern Med. 2010; 268(1): 15–24). Within this novel genus are newly discovered families of specialized pro-resolving mediators — including two coined the resolvins and protectins — which are generated during inflammatory resolution (Read, Specialized Pro-Resolving Mediators: A New Tool for Resolving Inflammation)
Mounting evidence shows that the various forms of resolvins, different types of which are synthesized from EPA and DHA, “possess potent anti-inflammatory and immunoregulatory actions.” Those actions include blocking the production of pro-inflammatory mediators, regulating the trafficking of white blood cells to inflammatory sites, and the clearance of neutrophils from mucosal surfaces. The protectins, also referred to as neuroprotectins, are synthesized specifically from DHA and promote brain health through anti-inflammatory and protective actions localized in the neural tissues.
Yanuck did point out one caviat to prescribing fish oil: “DHA in the presence of oxidative stress will turn into F2-isoprostanes, and those are nasty and inflammatory.”
Commonly, patients with autoimmune conditions exhibit high oxidative stress levels, so one needs to be careful when introducing fish oil supplements. “If you give fish oil to a patient with abundant oxidative stress, you’re gonna tank ’em,” Yanuck warned.
For these patients, he recommended instead using glutathione, N-acetyl cysteine (NAC), and acetylcysteine to quell the oxidative stress.
Living Off the Edge
In all autoimmune patients, a constellation of general triggers worsen the autoimmune processes. Factors like chronic psychosocial stress, infection, dysbiosis, dysglycemia, food reactions, impaired hepatic detoxification, environmental toxins, hormones, insomnia, and –in some cases–excessive fitness training, all shape the individual biological landscape and can push vulnerable patients off the edge and into the inflammatory ditch.
Any of the triggers on this list can initiate flares in complex autoimmune patients, says Yanuck. “If they get anemia? Ditch, flare. Infection? Flare. Insomnia? Flare. More stress from work? Flare. Hormonal problems? Flare. You must address the general triggers to move the patient further from the ditch.”
Even if you don’t fully understand a particular patient’s unique immunology, it makes sense to address these more general big picture triggers. It “gives you an advantage because, even before you have a lot of clarity about the case…you’ll move the person away from the ditch, no matter what autoimmune process it turns out that they have.”
The challenge for physicians is that, “you can’t micromanage the difference” between being on the cusp and falling into the ditch.
Patients “can’t succeed if they’re at the edge and live their lives that way, with the litmus test being, ‘as long as I’m not in the ditch, I’m okay, and if I do something that’s off-plan but I don’t fall in the ditch, that proves I’m okay.'”
This logic is dangerously faulty. “If they think that way, you may get them 50 yards from the ditch” to a place of relatively improved health. But some patients perceive as a green light to “cheat” on their treatment protocols. Because they feel good, they might, for instance, resume eating an offending food like gluten or dairy.
And that is a perfect way to land onesself squarely in the ditch…again.
A flaring patient might think, “I started doing that thing a month ago, so that can’t be it, because I was fine then…my flare is only two days old.”
Dr. Yanuck stressed the fallacy of that mode of thinking. In truth, that patient has likely been headed straight for the cliff for a while. Assuming no change in direction, the only difference between driving your car toward a cliff and driving your car off the cliff is time.
“The problem was, you aimed the car at the cliff and pressed the gas pedal.” But patients often do not understand that because they think in strict binary terms: “good” or “no good,” “safe or “not safe.” But the autoimmune process is much more of a continuum. “The edge of the ditch is part of the ‘no good.’ Fifty yards away from the is good.”
Epitope Spreading
There is another danger that autoimmune patients face if they repeatedly go off an effective therapeutic program: epitope spreading.
This is a process that scientists are still working to fully elucidate. It appears to stem from chronic immune responses that promote the development of new autoreactive B cell clones.
In a healthy body, the immune system uses B cells to make antibodies that protect us against specific antigens. In patients with autoimmune disease, this otherwise normal process becomes dysfunctional over time. The B-cells begin to produce antibodies that target not only non-self, but also self-antigens, attacking its own tissues and organs.
Every time a patient falls in the ditch of an autoimmune flare, Yanuck cautioned, they increase the likelihood of epitope spreading.
After a flare, a patient might expect that you can help them recover and return to a previous health baseline. E pitope spreading makes that much more difficult, if not impossible.
“Memory cells aren’t deletable — they’re always waiting,” explained Yanuck. The debris of tissue damage “gets taken up by antigen-presenting cells that go to the lymph nodes looking for a match.” If memory cells from a previous flare match self-tissue that is again presented as an antigen, the immune system creates new clones that target self-tissue, worsening the autoimmune flares.
It is very imprortant to educate patients about concepts like epitope spreading, so they understand the need to stick with a program that’s working and to avoid future flares at all costs. If they can understand a few basic immunological concepts, they’ll at least understand why they need to continue avoiding the laundry list of general triggers. They’ll also recognize that your recommendations are not meant to deprive them in any way, but that you are trying to lead them away from the pit of incessant flares and towards safer, healthier physiological ground.
END







