Daily supplementation with a fixed dose combination of nicotinamide riboside, L-carnitine, L-serine, and N-acetyl cysteine significantly reduced liver fat, quelled oxidative stress, and improved liver enzyme profiles in a cohort of 31 patients with non-alcoholic fatty liver disease (NAFLD).
Defined as hepatic fat accumulation of 5% or more, unrelated to alcohol consumption or other liver diseases, NAFLD affects roughly one in four adults according to recent estimates. Incidence is on the rise, driven in part by the close association of NAFLD with insulin resistance, diabetes, and obesity, all of which are increasing globally.
“Current management strategies, including lifestyle modification, increased physical activity, and dietary intervention, have limited adherence and minimal prolonged success,” wrote Mujdat Zeybel, of the NIHR Nottingham Biomedical Research Center, UK, and lead author of the new study.
“This study demonstrated CMCS supplementation significantly decreased liver fat, improved liver function, and decreased liver inflammation in NAFLD patients, suggesting CMCS may be an effective therapeutic strategy for NAFLD,”
Mujdat Zeybel, MD, NIHR Nottingham Biomedical Research Centre
“No drugs have been approved to treat NAFLD, and effective treatment options with long-term safety are urgently required.”
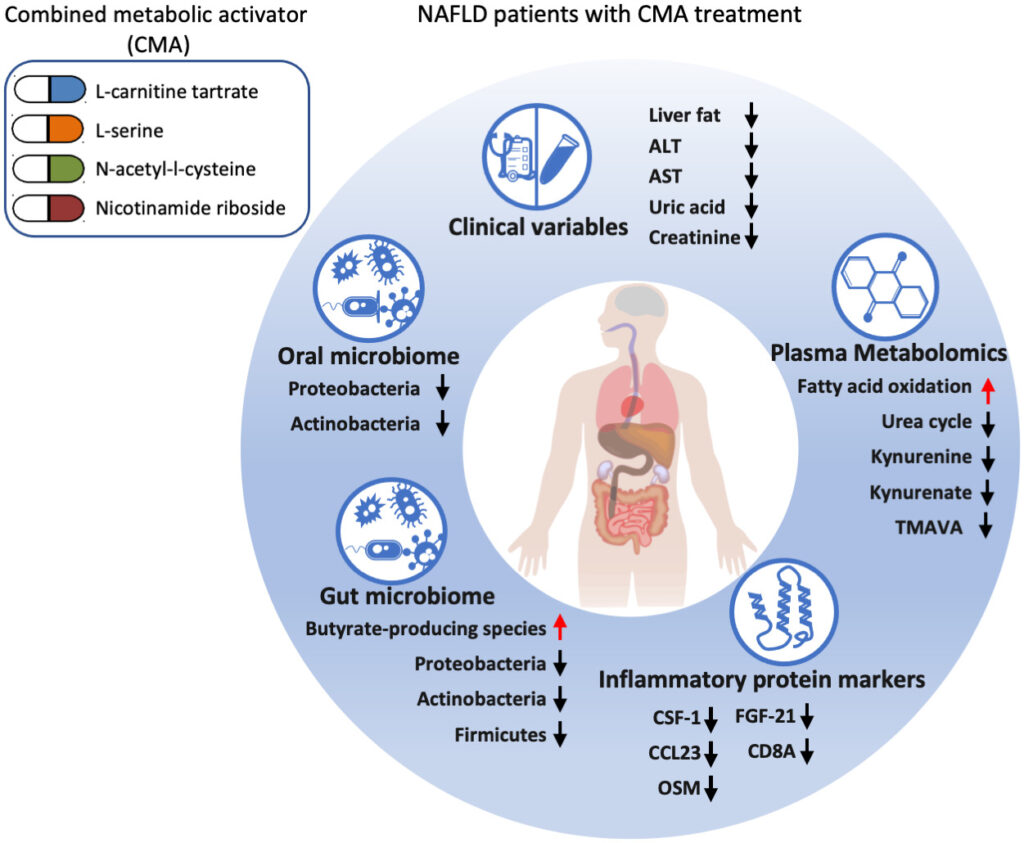
The combined metabolic cofactor supplementation (CMCS) regimen developed by the Nottingham group and used in this trial consists of:
- 1g nicotinamide riboside (NR), which raises mitochondrial NAD+ levels, and boosts fat metabolism
- 3.73g L-carnitine tartrate, which promotes transport of fat into the mitochondria
- 12.35g L-serine, which increases glutathione synthesis
- 2.55g N acetyl-L-cysteine (NAC), which also promotes glutathione production and reduces oxidative stress
A total of 31 patients with MRI-confirmed non-alcoholic steatosis were randomized to either placebo (N=11) or the CMCS combination (N= 20). They were instructed to take one dose of the assigned treatment after their evening meals for 14 days, and then two doses daily (after breakfast and dinner) for the next 56 days.
After 70 days of treatment, CMCS reduced hepatic fat as assessed by MRI by 10% compared with the placebo. There were other secondary improvements as well, including a 39% reduction in serum alanine transaminase (ALT), and a 30% drop in aspartate transaminase (AST), suggesting overall improvement in liver function. These changes were independent of weight loss (Zeybel M, et al. Mol Systems Biol. 2021).
The Nottingham researchers also saw significant drops in hepatic inflammatory mediators, a decrease in serum creatinine, and interestingly, a 5% decrease in systolic blood pressure among the CMCS patients.
Uric Acid Surprise
Notably, there was also a 12% decrease in uric acid associated with the supplement regimen. This, the authors note, was an unexpected but welcome surprise (read Dropping Acid with Dr. David Perlmutter).
“Uric acid stimulates hepatic steatosis either directly or by activating NLRP3 inflammasomes. Although the extent to which uric acid reduction contributed to the regression in hepatic steatosis is unclear, it likely had at least an additive effect,” wrote Zeybel.
Analysis of fecal and salivary samples showed beneficial microbiome changes among the CMCS-treated subjects, notably an increase in butyrate-producing organisms (Lawsonibacter asaccharolyticus, Eubacterium remulus and Eubacterium siraeum). “Butyrate is a preferred energy source for gut epithelial cells, and it plays a vital role in maintaining health in humans,” Zeybel points out.
The microbiome changes are important because NAFLD is associated with perturbations in the gut microbiome. Several studies have shown that people with NAFLD have an increased abundance of detrimental bugs like Actinobacteria and Proteobacteria. Obesity-related NAFLD is associated with an increased ratio of Firmicutes to Bacteroidetes in the gut.
Three Landmark Features
The rationale for developing this multimodal supplement combination emerged from the Nottingham group’s previous work which identified, “three landmark metabolic features of hepatic steatosis: limited serine availability, reduced de novo glutathione (GSH) synthesis, and altered nicotinamide adenine dinucleotide (NAD+) metabolism.”
They hypothesized that supplementation with L-carnitine tartrate would facilitate mitochondrial fatty acid uptake from cytosol, while the NAD+ precursor nicotinamide riboside would induce hepatic mitochondrial β-oxidation and facilitate mitochondrial fatty acid transfer. L-serine and NAC, which are key glutathione precursors, would help to reduce oxidative stress.
“We further hypothesized that supplementation with these metabolic cofactors would promote mitochondrial fatty acid uptake and oxidation, and reduce hepatic fat and inflammation,” Zeybel said, adding that the underlying mechanism in the pathogenesis of NAFLD is inadequate removal of hepatic fat by fatty acid oxidation.
Preclinical studies showed that indeed, the Nottingham group was on the right track. The four combined metabolic cofactors did appear to promote the oxidation of fat, reduce oxidative stress, activate mitochondria, and facilitate the transfer fat out of the liver. Those earlier findings justified further human clinical studies.
“The results of this study are consistent with the results of the phase 1 pilot clinical study showing CMCS supplementation was safe and well-tolerated in patients with NAFLD, and highlights the important role of mitochondrial health. This study demonstrated CMCS supplementation significantly decreased liver fat, improved liver function, and decreased liver inflammation in NAFLD patients, suggesting CMCS may be an effective therapeutic strategy for NAFLD,” the authors conclude.
-END-







