Among the core tenets of holistic and functional medicine are the ideas that lifestyle-based interventions aimed at mitigating root causes of chronic disease will improve clinical outcomes, and that this can reduce healthcare costs.
These premises makes intuitive sense. But proving them on a healthcare system level has been challenging.
Two recent studies provide significant new evidence to support these concepts.
Setting Back the Clock
According to Kara Fitzgerald and colleagues, a comprehensive lifestyle change program centered on a plant-based, low-carb, high protein diet, use of probiotics, daily exercise, and regular stress management, can reverse genome-wide DNA changes associated with aging and disease risk. In essence, it can set the biological clock back by as much as 2-3 years, in a relatively short period of time.
Fitzgerald, a naturopathic doctor practicing functional medicine in Sandy Hook, CT, collaborated with colleagues at the National University of Natural Medicine, the American Nutrition Association, McGill University, and the University of California, San Diego, to study the impact of this multimodal intervention on biochemical and genetic markers of aging in a cohort of 38 generally healthy middle-aged men.
Compared with the 20 men in the control group, who did not undertake any type of lifestyle change, those in the program showed a 3.23 year decrease in their DNAmAge after 8 weeks.This difference was statistically significant.
Following a 3-week washout period of abstention from alcohol, nicotine, cannabis, or other recreational drugs, and discontinuation of any nutrition supplements or herbs beyond daily basics like multivitamins, vitamin D, and fish oils, the participants were randomized to the lifestyle intervention (N=22) or to a control group (N=22) that had no intervention.
The lifestyle change program included:
- Coaching for implementation of a plant-centered, low-carb, high-protein diet rich in foods that provide methylation substrates and cofactors (eg, folate, betaine); ten-eleven translocation (TET) demethylase cofactors like alpha ketoglutarate, vitamin C, and vitamin A; and polyphenols (curcumin, EGCG, rosmarinic acid, quercetin, luteolin).
- Daily supplementation with a polyphenol-rich fruit and vegetable powder (Phytoganix by Metagenics), and a probiotic providing 40 million CFU of Lactobacillus plantarum 299v (Ultraflora Intensive Care by Metagenics).
- A minimum of 30 minutes of exercise per day, at least 5 days per week, at an intensity of 60-80% of maximum perceived exertion.
- Twice-daily breathing exercise sessions to elicit the Relaxation Response, based on techiques developed by stress research pioneer Herbert Benson, MD.
Participants were also encouraged to optimize their sleep, with a recommendation for at least seven hours nightly.
“Methylaging”
The study was designed to assess the degree to which the intervention could change participants’ physiologic age as estimated by the DNAmAge clock.
This tool calculates aging based on changes in DNA methylation– the addition or subtraction of methyl groups to cytosine residues at specific points on chromosomes. Methylation is a key mechanism for controlling gene expression. There are more than 20 million methylation sites on the human genome, a few thousand of which correlate strongly with aging.
According to Fitzgerald and colleagues, “the best biochemical markers of an individual’s age are all based on patterns of methylation.” They can be easily and inexpensively measured in tissue or saliva samples.
“As of this writing, the best-studied methylation-based clock is the multi-tissue DNAmAge clock,” she notes. “Horvath’s DNAmAge clock predicts all-cause mortality and multiple morbidities better than chronological age.”
Developed by geneticist Steve Horvath at the University of California Los Angeles, the DNAmAge calculator, like other methylation clocks, systematically quantifies and analyzes the methylation changes that occur as people grow older. The actual testing method—the Illumina Methylation Epic Array—uses buccal cells and white blood cells from saliva samples. This makes it a simple, non-invasive, and patient-friendly assessment tool.
Horvath’s research has shown that roughly 60% of CpG sites in the human genome lose methylation with age, while 40% gain methylation.
This is an important point: it is the pattern of methylation–not the total amount—that matters in assessing age and disease risk. Fitzgerald stressed that in her study, “a net increase in methylation is not the therapeutic target.” Rather, the lifestyle program was designed to foster a healthier, more youthful pattern of methylation and demethylation.
“We have tentatively accepted the hypothesis that the methylation pattern from which the DNAmAge clock is computed is a driver of aging (and the chronic diseases of aging), thus we expect that attempting to directly influence the DNA methylome using diet and lifestyle to set back DNAmAge will lead to a healthier, more “youthful” metabolism.”
Prior studies suggest that this is a reasonable hypothesis.
A 2017 paper by Quatch and colleagues at UCLA showed a modest but significant reduction in DNAmAge in individuals keeping a lean meat, fish and plant-based diet (Quatch A, et al. Aging. 2017).
The dietary guidelines in the Fitzgerald study raise the intake of nutrients that are substrates or cofactors in methylation biosynthetic pathways. The L. plantarum probiotic supplement was included because this organism is known to be a folate producer.
“The best biochemical markers of an individual’s age are all based on patterns of methylation. It is the pattern of methylation–not the total amount—that matters in assessing age and disease risk.”
– Kara Fitzgerald, ND
Stress, Methylation, & Aging
There’s good evidence that certain forms of exercise will positively impact methylation patterns. For example, in a 2012 study of 500 Australian women, regular tai chi practice was associated with slowing of age-related DNA methylation losses (Ren H, et al. Ev Based Compl Altern Med. 2012). A study of 647 women by University of North Carolina researchers showed that those who had a lifelong history of exercise had DNA methylation patterns consistent with reduced cancer risk (White AJ, et al. Eur J Cancer. 2013).
Emotional stress is a major influence on age-associated DNA methylation patterns. Fitzgerald and colleagues point out that roughly one quarter of all DNAmAge CpG sites are located in glucocorticoid response elements within the genome, strongly suggesting a relationship between stress and accelerated aging. In 2015, Zannas and colleagues reported a clear association between cumulative lifetime stress and accelerated aging of the methylome (Zannas AS, et al. Genome Biology. 2015).

The good news is that this is modifiable. Investigators at the University of Padua showed that twice-daily practice of the Relaxation Response for 60 days could markedly reduce DNAmAge in healthy women and men, independent of any other medical intervention (Pavanello S, et al. Int J Environ Res Publ Health. 2019)
Metabolic Reset
In Dr. Fitzgerald’s study, published in the journal Aging, 18 of the 22 subjects assigned to the diet and lifestyle treatment arm completed the full 8-week intervention.
Compared with the 20 men in the control group, who did not undertake any type of lifestyle change, those in the program showed a 3.23 year decrease in their DNAmAge (p=0.018) after 8 weeks.This difference was statistically significant.
DNAmAge of those in the treatment group decreased by an average 1.96 years. In contrast, the men in the control group scored an average of 1.27 years older at the end of the study period compared to their baseline scores, though this difference was not statistically significant (p= 0.153).
The men following the diet and lifestyle protocol also showed meaningful changes in mean serum 5-methyltetrahydrofolate, which increased from an average of 78 to 88 nmol/L (+15%, p=0.004). Mean triglycerides dropped from an average of 112 to 89 mg/dL (-25%, p=0.009). These findings are not surprising, given that the recommended diet was folate rich, and limited in saturated fat intake.
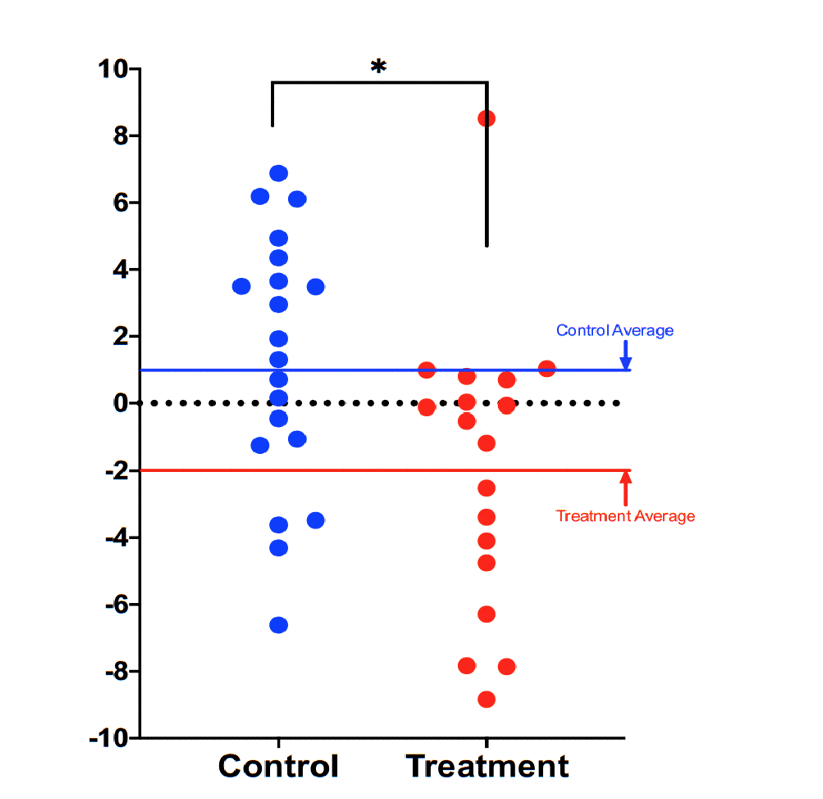
The lifestyle participants also showed significant decreases from their baseline measures of total cholesterol (-22.8 mg/dL, p=0.004) and LDL cholesterol (-16.8 mg/dL, p=0.01).
There were no major differences between the two groups on risk factors such as blood glucose, hemoglobin A1C, total cholesterol, HDL cholesterol, LDL cholesterol, methionine, s-adenosylmethionine (SAM), s-adenosylhomocysteine (SAH), or the SAM:SAH ratio.
The authors point out that all participants were generally healthy, and without major disease risk profiles. So major differences on these cardiometabolic variables were not expected.
The key point is that a relatively brief but comprehensive lifestyle shift could produce a measurable and meaningful change in age-related gene expression.
“To our knowledge, this is the first randomized controlled study to suggest that specific diet and lifestyle interventions may reverse Horvath DNAmAge epigenetic aging in healthy adult males,” the authors conclude (Fitzgerald KN, et al. Aging. 2021). The study was funded by an unrestricted grant from Metagenics.
A Starting Point
Fitzgerald and colleagues acknowledge that this study has limitations, and that it is a starting point rather than a final word on the subject.
The sample size was small, and while the DNAmAge changes do suggest a favorable shift toward reduced risk of cardiometabolic disease, it is difficult to draw firm conclusions about disease risk reduction, given the brief 8-week intervention period and the general health of the study population.
Further, none of the methylation clock calculators currently in use have been statistically validated as predictors of disease risk.
“A multi-modal lifestyle shift has direct and measurable impact on the epigenetics of aging.”
It is not possible to determine from the available data whether the DNAmAge reversal observed in the lifestyle change cohort would persist if—or perhaps we should say when–participants return to their previous pre-intervention habits.
Lack of diversity is an issue, too. There were only 2 African-Americans and 4 Asian-Americans in the study cohort, making it difficult to generalize the findings to populations beyond middle-aged white males of European descent.
The authors explain that they focused on the 50-72 years-old age bracket because this is the time when age-associated vulnerabilities typically manifest. Their decision to limit participation to men was, “to avoid the potential confounding factor of pre-, peri-, and post-menopausal sex hormone levels of the same age range in women.”
But these limitations do not invalidate the main finding, which is that a multi-modal lifestyle shift has direct and measurable impact on the epigenetics of aging. The authors believe this has public health implications.
“Advanced age is the largest risk factor for impaired mental and physical function and many non-communicable diseases including cancer, neurodegeneration, type 2 diabetes, and cardiovascular disease,” they write.
“Considering economics alone, delaying aging by 2.2 years (with associated extension of healthspan) could save $7 trillion over fifty years. This broad approach was identified to be a much better investment than disease-specific spending. Thus, if interventions can be identified that extend healthspan even modestly, benefits for public health and healthcare economics will be substantial.”
The Power of Group Visits
Researchers at the Cleveland Clinic Center for Functional Medicine have published a new study validating both the clinical and economic value of shared medical appointments (SMA), also known as group visits, for improving physical and mental health of people with a variety of chronic disorders.
Michelle Beidelschies, PhD, and colleagues looked at outcomes and healthcare costs for 2,455 patients receiving functional medicine care at the Cleveland Clinic. The vast majority—2,229 patients—obtained the clinical services via traditional individual visits, while 226 participated in SMA programs. They sought functional medicine at Cleveland Clinic for a range of chronic conditions and concerns.
The multidisciplinary SMA programs, called Functioning For Life® (FFL), were designed around five general clinical foci: Weight management (36% of patients); Autoimmune disorders (34%); Digestive disorders (17%); Women’s health (8%); and Diabetes (5%).
FFLs are all 10-week programs that emphasize food as medicine, with specific recommendations aimed at remedying the conditions in question. As is the case with functional medicine in a one-on-one setting, the FFL model takes a root-cause approach to identifying and reducing triggers for illness—poor diet, stress, toxins, allergens, infections, dysbiosis, and genetic factors—while simultaneously optimizing factors that engender good health.
Cleveland Clinic’s SMAs are headed by multidisciplinary teams. Four sessions are co-led by an MD, DO, Nurse Practitioner, or Physician Assistant, along with a health coach, and six sessions are led by a registered dietitian.
Previous studies suggest that the SMA model is able to improve clinical outcomes in people with type 2 diabetes, obesity, and chronic pain syndromes, and that functional medicine carried out in a group visit setting could be more cost-effective than in the standard individual visit framework.
A PROMISing Approach
Using the Patient-Reported Outcomes Measurement Information System (PROMIS) to assess clinical outcomes, Dr. Beidelschies and her team sought to compare functional medicine in the SMA context head to head with similarly targeted services in the individual setting. The primary endpoint was the change in PROMIS GPH at 3 months. Secondary outcomes included change in PROMIS Global Mental Health (GMH), biometrics and cost.
Simply put, SMAs are a more efficient way to provide functional medicine services, at least for certain types of patients with certain kinds of conditions.
The two cohorts (individual and SMA) were matched on age, gender, marital status, income, body mass index, blood pressure, baseline PROMIS scores, and diagnostic category.
Compared with propensity-matched patients in the individual settings, those in the SMA programs showed significantly greater improvements in their PROMIS Global Physical Health (3.3 vs 2.1 points) and Global Mental Health (3.1 vs 1.3) scores by the end of the third month (Beidelschies M, et al. BMJ Open. 2021).
Nearly 40% of the SMA patients had improvements of 5 or more points on their PROMIS scores, versus 28% in the individual setting—an interesting but not statistically significant difference.
The group visit patients lost more weight, with an average decrease of 3.8 kg versus 2.4 kg in the individual visit cohort, equating to a mean weight loss of 4.4% of total body mass versus 2.8%. Both groups showed similar improvements in systolic and diastolic blood pressure.
More Touchpoints, Greater Savings
The SMA model provides patients with a greater number of visits than the standard individual visit model. So long as there are 6 or more patients per group, it costs less to deliver, and generates greater revenue per patient for the clinic.

Beidelschies and colleagues report that in the Cleveland Clinic study, the SMA patients averaged roughly 9 visits per patient during the 3-month study period, versus an average of 2 visits per patient in the individual setting. The group setting also provided far more caregiver time and attention (14 hours versus 3.5 hours).
Patients in Cleveland Clinic’s SMA programs pay a fixed cost of $1549, which proved to be roughly 5% lower than the average per-patient cost of $1633 for individual visits during that same 3-month period.
So long as there are 6 or more patients per group, it costs less to deliver, and generates greater revenue per patient for the clinic.
– Michelle Beidelschies, PhD
In total, the SMA model generated 11% more revenue per patient than the individual visit setting ($4204 vs $3780). In the Cleveland Clinic model, an SMA reaches the break-even point with 3 patients enrolled.
This is not the first study to demonstrate the clinical value of SMAs. Dr. Beidelschies notes that “Studies of SMAs for pre-diabetes and weight management have found that patients in SMAs lost at least twice as much weight as those with individual visits. Cole et al reported patients participating in a pre-diabetes nutrition SMA lost 3.5% of their body weight at 3 months compared with 1.8% in an individual setting.”
Simply put, SMAs are a more efficient way to provide functional medicine services, at least for certain types of patients with certain kinds of conditions. It offers many advantages including:
- More practitioner-patient touch points
- Greater diversity of expertise
- Peer to peer learning and support
- Opportunities for in depth education on nutrition and cooking
- Stronger therapeutic alliances
- Goal-setting and accountability
- Regularly scheduled visits obviate the often haphazard appointment process common to many individual visit settings
“This study is a first-of-its-kind to demonstrate that functional medicine-based care can be efficiently delivered in a shared medical appointment setting and contribute to improved patient outcomes even beyond those achieved in an individual appointment setting. This innovative and transformative model redesigns the way we think about conventional healthcare delivery,” Dr. Beidelschies writes.
END







