 Chronic kidney disease (CKD) is seldom discussed in integrative health care circles. Perhaps this is because it is often silent, progressing insidiously alongside other interrelated chronic conditions that receive more attention such as diabetes, hypertension, and cardiovascular disease.
Chronic kidney disease (CKD) is seldom discussed in integrative health care circles. Perhaps this is because it is often silent, progressing insidiously alongside other interrelated chronic conditions that receive more attention such as diabetes, hypertension, and cardiovascular disease.
But it is a condition that warrants our clinical attention, firstly because it is very common, and secondly because there’s quite a lot we, as holistic practitioners, can do to help avert the devastating late stage consequences of kidney disease.
CKD typically advances over many years, with a long latent period when the disease is asymptomatic and more or less invisible. The advancing age of our population and the increased prevalence of chronic inflammatory conditions mean that CKD will be increasingly prevalent in the coming years. Integrative practitioners can provide critical screening, early identification, and natural preventive interventions for our elders.
For those already living with CKD, holistic interventions aimed at supporting the kidneys can limit risk, slow progression, and improve both quality of life and lifespan.
New and exciting findings are emerging from the field of nephrology research indicating that the intestinal microbiota and gut barrier function play key roles in the development and progression of CKD. Like most organ systems, the renal system is affected by what goes on in the gut. Microbiome imbalances or the presence of particular organisms in the GI tract can have direct and indirect detrimental effects on the kidneys.
A Silent Epidemic
CKD is already reaching epidemic proportions in the US and worldwide. According to the National Kidney Foundation, one out of every three American adults is at risk, and 30 million already have some level of CKD. Though it is often thought to be a result of cardiovascular disease, the statistics show that the relationship is actually bi-directional: presence of CKD is highly linked with increased cardiovascular risk, as well as with cognitive impairment, and increased mortality from all causes.
When it comes to the kidneys, prevention and preservation of function are paramount.
The initial stages of CKD can be managed to limit progression, but unfortunately, once the disease advances beyond a certain point, end stage renal disease (ESDR) requiring dialysis is all but inevitable (Kittiskulnam P. et al. Semin Nephrol. 2016 Jul;36(4):305-18 ).
High risk groups include people with diabetes, hypertension and other cardiovascular conditions. Risk is also elevated among certain ethnic groups, especially African Americans, Hispanics, Pacific Islanders, and Native Americans.
Do any of your patients have genetic, autoimmune, and obstructive kidney conditions, or repeated UTI’s? If so, they’re at risk, regardless of age or ethnicity.
Also keep in mind that OTC drugs such as non-steroidal anti-inflammatory drugs (NSAIDs) — acetaminophen and aspirin – can raise the risk. So do some prescription antibiotics, antihypertensive, antiviral, and ulcer medication. Prescription laxatives, chemotherapy drugs and contrast media, are also bad news for the kidneys.
Then, there are the illicit drugs including cocaine, heroin, and amphetamines that can also injure kidney tissue and increase risk of CKD.
Given how common are these risk factors, we need to keep the index of suspicion for CKD very high.
CKD Pathogenesis
The kidneys play many essential physiological roles; In addition to the obvious blood filtering and electrolyte balancing functions, they’re involved in the regulation of hormones that impact bone health, hematopoiesis, and blood pressure.
Those who practice Traditional Chinese Medicine (TCM) have a sophisticated understanding of the many aspects of kidney function, some of which are only now being corroborated by modern research.
In TCM theory, the kidneys hold the “essence” (“jing”) of vitality, both inherited and current, having both yin and yang aspects  to influence physical reproduction, development, fluid balance, bone and blood production. They also influence subtler cognitive and emotional aspects influencing memory and will power.
to influence physical reproduction, development, fluid balance, bone and blood production. They also influence subtler cognitive and emotional aspects influencing memory and will power.
The ancient Chinese understood that kidney functions are central to overall health; if the kidneys don’t function well, neither does anything else.
Kidney damage is the result of an inflammatory response that is triggered when the kidneys attempt to repair themselves from some sort of insult or injury. When there is ongoing or repeated insult, the inflammation becomes chronic.
Degradation of the kidney results in a decrease in the estimated glomerular filtration rate (eGFR). This is due to the formation of scar tissue (fibrosis) and loss of tissue architecture. If this progresses far enough, the organ simply cannot function (Henderson NC, et al. Am J Pathol. 2008;172(2):288-98).
The Gut-Kidney Connection
An abnormal intestinal microbiome contributes to the accumulation of gut derived uremic toxins, and high circulating levels of bacteria-derived lipopolysaccharides (LPS), both of which can be nephrotoxic. It also leads to immune dysregulation. All of these are factors in the pathogenesis of CKD. (Pan W, et al. Int Urol Nephrol. 2017 Aug 28).
An altered microbiome also fosters the development of a chronic systemic inflammatory state which is associated with a plethora of comorbidities, including obesity, insulin resistance, diabetes, and CVD. This creates a domino effect of interrelated conditions all impacting the kidneys (Mafra D, et al. Future Microbiol. 2014;9(3):399-410).
CKD can be triggered by endotoxins from gut bacteria, such as metabolites from protein fermentation. These toxins can provoke a powerful inflammatory response that can be very damaging to the kidneys. Disruption of gut barrier function (aka “leaky gut”) allows translocation of these endotoxins and bacterial metabolites to the systemic circulation, which contributes to uremic toxicity, inflammation, progression of CKD, and CVD.
There’s some evidence that targeted pre- and probiotic interventions to re-establish intestinal symbiosis, neutralize bacterial endotoxins, or adsorb gut-derived uremic toxins, can slow progression of CKD (Ramezani A, et al. J Am Soc Nephrol. 2014 Apr;25(4):657-70).
Many integrative therapies are adept at treating dysbiosis and “leaky gut”. We have a well-established toolbox in hand for doing so.
Gal-3: The Orchestrator of Fibrosis
Another promising area of investigation centers around our understanding of how fibrosis develops, which can translate into how we assess and then interrupt this process prior to irreversible damage.
A protein called galectin-3 (Gal-3) is a key driver of the fibrotic process in the kidneys, as well as other vital organs including the liver and heart.
Gal-3 is a beta-galactoside binding protein, that binds to glycoprotein and glycolipids at its carbohydrate recognition domain (CRD). Gal-3 performs essential functions inside cells, among which is the regulation of cell cycling. It has multiple roles that are tissue and location-dependent.
Once Gal-3 is upregulated in response to injury or inflammation, it is excreted out of the cell into the extracellular matrix and the circulation, where it plays a key role in collagen synthesis to repair and remodel tissue. Normally, in response to discrete events, this is a self-limiting process. But like inflammation, it can easily go awry.
When Gal-3 is upregulated in response to repeated injury or chronic inflammation, it is continually excreted to the extracellular spaces and ultimately into the circulation where it drives further inflammation and fibrosis in vital organs. It also disrupts cell-cell communication, interferes with immune competence, and promotes cancer growth, invasion, and metastasis (Fortuna-Costa A. et al. Front Oncol. 2014 Jun 16;4:138).
The kidneys provide a perfect illustration of both the normal and pathogenic roles of Gal-3.
High levels of intracellular Gal-3 are vital for development and growth of nephrons. But in the adult kidney, Gal-3 levels are normally low (Chen SC, et al. Int J Mol Sci. 2016 Apr 14;17(4):565). When a kidney is injured, Gal-3 production surges and is excreted outside the cell as part of the repair mechanism.
When this becomes chronic, the excess Gal-3 activates interstitial cells (macrophages, fibroblasts, and myofibroblasts) and secretes other fibrotic molecules such as TGF-β1 and IL-1β, to produce collagen ultimately leading to tissue remodeling and fibrosis.
Numerous studies show that Gal-3 is upregulated in CKD, and that higher serum levels of Gal-3 correlate to more advanced stages (Desmedt V, et al. Am J Nephrol. 2016;43(5):305-17. Drechsler C, et al. J Am Soc Nephrol. 2015 Sep;26(9):2213-21).
MCP: A Natural Gal-3 Inhibitor
Fortunately, there is a natural Gal-3 inhibitor–modified citrus pectin (MCP)–which has been well researched for its effectiveness in blocking Gal-3 activity and promoting kidney health.
 MCP is derived from the inner pith of citrus peels. A number of studies show that it is a natural binder and inhibitor of Gal-3. The benefits of Gal-3 inhibition using MCP have been shown in studies of patients with progressive fibrotic conditions, including CVD, liver fibrosis, CKD, and cancer. (Nangia-Makker P, et al. J Natl Cancer Inst. 2002 Dec 18;94(24):1854-62, Martínez-Martínez E, et al. Hypertension. 2015 Oct;66(4):767-75, Glinsky VV, et al. Carbohydr Res. 2009 Sep 28;344(14):1788-91).
MCP is derived from the inner pith of citrus peels. A number of studies show that it is a natural binder and inhibitor of Gal-3. The benefits of Gal-3 inhibition using MCP have been shown in studies of patients with progressive fibrotic conditions, including CVD, liver fibrosis, CKD, and cancer. (Nangia-Makker P, et al. J Natl Cancer Inst. 2002 Dec 18;94(24):1854-62, Martínez-Martínez E, et al. Hypertension. 2015 Oct;66(4):767-75, Glinsky VV, et al. Carbohydr Res. 2009 Sep 28;344(14):1788-91).
PectaSol-C® Modified Citrus Pectin is the only available patented form of MCP supported by extensive research.
The role of Gal-3 in health and disease, and the use of MCP to inhibit Gal-3 when it is pathogenic have been a primary focus of my research efforts for the past 20 years. I also routinely apply this Gal-3 inhibition strategy in clinical practice.
I am certainly not alone in this. Many independent researchers are also studying PectaSol-C MCP. The data from both preclinical and human clinical studies is quite strong (Yu L. Circ Heart Fail. 2013 Jan;6(1):107-17, Abu-Elsaad NM. et al. Can J Physiol Pharmacol. 2016 May;94(5):554-62, Kolatsi-Joannou M. PLoS One. 2011 Apr 8;6(4):e18683).
With no known side effects and a GRAS (generally recognized as safe) designation, MCP is clearly a powerful tool for a proactive, integrative approach to kidney health.
Assessing CKD Risk
The first step in preventing or slowing CKD is to make a thorough assessment of kidney function.
Standard renal function tests are well established: creatinine, Blood Urea Nitrogen (BUN), BUN-creatinine ratio, eGFR, and 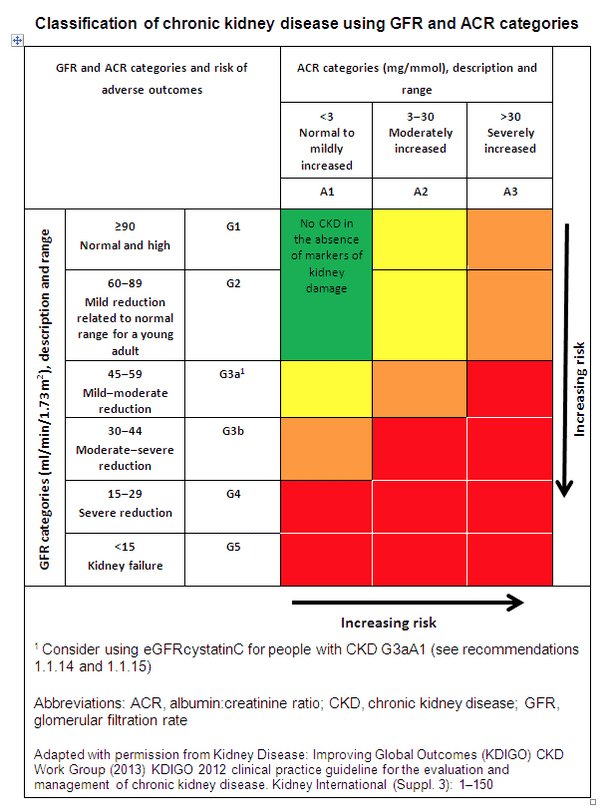 urine albumin or the albumin:creatinine ratio (ACR). These measurements will give a fairly accurate picture of how well a patient’s kidneys are doing their various physiological jobs.
urine albumin or the albumin:creatinine ratio (ACR). These measurements will give a fairly accurate picture of how well a patient’s kidneys are doing their various physiological jobs.
CKD staging is based on two biomarkers: eGFR and ACR. Albuminuria is one of the most prognostic biomarkers (van der Velde M, et al. Kidney Int. 2011; 79:1341–52). Across every stratum of eGFR, elevated amounts of albumin in the urine are associated with increased risk of CVD, CDK progression, and death (Levey AS, et al. Kidney Int. 2011;80(1):17-28).
The Renal Association, based in the UK, has published detailed information about CKD staging categories.
In practice, however, we need to be careful with staging criteria based heavily on serum creatinine, because low muscle mass (sarcopenia)—which is very common in elderly people–influences creatinine levels, but may not always be associated with advanced CKD.
Newer biomarkers are being researched to improve diagnostic accuracy. Cystatin-C is one example. Tests for this marker are now available. Unlike creatinine, cystatin-C appears to be independent of age, gender, and muscle mass. For a detailed review of emerging biomarkers see (Ferguson MA. Clin Chem. 2012 Apr;58(4):680-9).
There’s merit to the TCM view that the kidneys are far more than just passive blood filters and fluid regulators. If we’re going to take a truly holistic approach to preventing CKD, we need to look beyond the kidneys themselves.
I recommend a thorough terrain assessment including metabolic and inflammatory biomarkers, (Gal-3), evaluation of intestinal barrier function, microbial balance and oxidative stress biomarkers. Keep an eye open for indicators of metabolic syndrome, insulin resistance, hypertension, and cardiovascular comorbidities–all will have an adverse impact on kidney health.
Always ask patients about their prescriptions and OTC drug use. It’s probably a good idea to ask about supplements, too. And don’t forget about “recreational” drugs and drugs of abuse. That’s a sensitive topic, but it’s important. All of them can potentially contribute to CKD.
Preventing and reducing chronic inflammation is a critical aspect of minimizing CKD risk. An anti-inflammatory diet, moderate exercise program, and attention to stress reduction all work to decrease the pro-inflammatory milieu. Protocols should include botanical and nutrient support for intestinal barrier and gut microbial.
Kidney-targeted botanicals include Astragalus (Wang H, et al. Life Sci. 2004 Feb 13;74(13):1645-58) and Milk Thistle/silymarin (Vessal g, et al. Ren Fail. 2010 Jul;32(6):733-9). Supplements like pyridoxyl-5 phosphate, CoQ10, alpha lipoic acid, vitamins C, E, and folate are also helpful. All of these have protective effects against oxidative stress.
With a toolbox of natural solutions backed by the latest research, integrative practitioners are well positioned to provide critical interventions to stem the emerging epidemic of CKD.
END
Isaac Eliaz, MD, is an integrative medical doctor, licensed acupuncturist, researcher, product formulator and frequent lecturer. He has been a pioneer in holistic medicine since the early 1980s, and has published numerous peer-reviewed research papers. He is founder and medical director of the Amitabha Medical Clinic in Santa Rosa, CA, an integrative center specializing in cancer, Lyme disease, and other chronic conditions. He is also the founder and chief formulator of Clinical Synergy, a company providing targeted, research based, physician-formulated nutraceuticals.







