 Nutritional status is a major determinant of COVID-19 risk. Yet public health policy worldwide has given little attention to the role that nutrition, supplementation, and herbal medicine could play in improving individual and community resilience.
Nutritional status is a major determinant of COVID-19 risk. Yet public health policy worldwide has given little attention to the role that nutrition, supplementation, and herbal medicine could play in improving individual and community resilience.
The intense focus on stopping viral spread is sensible and necessary. But without a parallel effort toward strengthening immune system function, enhancing resilience, and mitigating the burden of chronic metabolic disease, people will remain vulnerable to SARS-CoV-2, and any future novel pathogen.
Pharmaceuticals and vaccines—the great hopes of political leaders, and of many citizens—will play important roles in quelling this pandemic. But they are only partial solutions—and they are still a long way off.
“A wealth of mechanistic and clinical data show that vitamins, including vitamins A,
B6, B12, C, D, E, and folate; trace elements, including zinc, iron, selenium, magnesium, and copper; and the omega-3 fatty acids EPA and DHA acid play important and complementary roles in supporting both the innate and adaptive immune system,” writes UK researcher Philip C. Calder in a recent paper in the journal, Nutrients.
“Deficiencies or suboptimal status in micronutrients negatively affect immune function and can decrease resistance to infections….there can be decreases in the numbers of lymphocytes, impairment of phagocytosis and microbial killing by innate immune cells, altered production of cytokines, reduced antibody responses, and even impairments in wound healing.”
Yet none of this is even part of the international public health dialog. The World Health Organization’s massive multi-nation SOLIDARITY Trial of the “most promising therapies” for COVID includes nothing about nutritional supplementation. The study is looking at the newly-approved antiviral Remdesivir, Chloroquine, Hydroxychloroquine, and a combination of two anti-HIV drugs—lopinavir and ritonavir.
In the absence of definitive drug therapies, practitioners are experimenting with a wide range of supplements, herbal remedies and older, rarely used drugs to try and protect themselves and help their patients deal with COVID.
Here, we review the natural therapies gaining the attention of prominent holistic and functional medicine practitioners and organizations. This list is by no means exhaustive, and the evidence is drawn mostly from studies of acute respiratory distress syndrome (ARDS), and other viral and bacterial respiratory infections. Given that COVID-19 did not even exist 6 months ago, there are few nutritional intervention studies in COVID patients, though several are in the works.
The Institute for Functional Medicine’s COVID-19 Task Force published a useful compendium of recommendations for nutrients and herbs that may:
- Favorably modulate viral-induced pathological cellular processes.
- Promote viral eradication or inactivation.
- Mitigate collateral damage from other therapeutic agents.
- Promote resolution of collateral damage and restoration of function.
- Reduce severity and duration of acute symptoms.
- Support recovery and reduce long-term morbidity and sequelae.
The functional medicine approach is focused, in part, on reducing the virus’ characteristic activation of a branch of the immune system called the NLRP3 inflammasome.
VITAMIN C
Vitamin C supports immune function in multiple ways: it strengthens epithelial barrier function, fosters growth and function of innate and adaptive immune cells, facilitates white blood cell migration to sites of infection, and increases phagocytosis and production of antibodies.
 Deficiency is clearly associated with increased susceptibility to severe respiratory
Deficiency is clearly associated with increased susceptibility to severe respiratory
Infections (Carr AC, Maggini S. Nutrients 2017). A Cochrane metanalysis of three small studies suggests that Vitamin C supplementation can reduce risk of pneumonia; the effect seems most pronounced in the elderly and those who are deficient at baseline (Hemila H, et al. Cochrane Database Syst Rev 2013). It also reduces duration and severity of upper respiratory tract infections (Hemila H, Chalker E. Cochrane Database Syst Rev. 2013)
The recent Calder paper recommends daily supplemental doses of at least 200 mg/day for healthy people, and between 1-2 g/day for those who are sick. These levels are considerably higher than the US RDAs, which are 75 mg/day for women and 90 mg/day for men, but still within the tolerable upper limit, which is 2 grams.
IFM’s guidance document suggests going up to 3 grams in high-risk individuals. The potential benefits are high, and the risk is very low.
The only COVID-specific studies have been with intravenous ascorbic acid. Data from COVID wards in Shanghai indicate that IV injections of 100-200 mg/kg per day improved oxygenation, reduced mortality, and shortened hospitalizations in COVID patients under ICU care. It also mitigates the characteristic cytokine storms seen in severe COVID cases. Expert guidelines from Shanghai advise giving the IV ascorbate in conjunction with heparin and broad-spectrum protease inhibitors.
The CITRIS-ALI (Vitamin C Infusion for Treatment in Sepsis Induced Acute Lung Injury)
study set the critical care world on fire when it published last January. The study involved 167 ARDS inpatients randomized to IV vitamin C (50mg/kg every 6 hours for 96 hours) or placebo infusion.
The vitamin C infusion did not result in any changes in organ failure scores, c-reactive protein levels, or any of the other primary endpoints. But it did produce a marked reduction in 28-day mortality (30% vs 44%), a greater number of ICU-free days (11 vs 8), and an increased number of hospital-free days (23 vs 16). These findings were statistically significant.
Yet, US hospitals have been slow to embrace IV ascorbate. The Front Line COVID-19 Critical Care Working Group hopes to change that. This physicians’ consortium from critical care centers all over the US advocates a protocol it calls MATH (Methylprednisolone, Ascorbic Acid, Heparin, and Thiamine).
The group recommends IV ascorbate, 3 grams per 100 ml every 6 hours, for 7 days, to counteract the inflammatory damage, and the severe hypercoagulable states typical of COVID. They contend that if implemented early, MATH could save thousands of lives, reduce the need for intensive care and mechanical ventilation.
VITAMIN D
Vitamin D and its metabolites are, in essence, immunoregulatory hormones. They have direct antimicrobial effects and indirect stimulatory effects on various aspects of the immune system. Calder points out that many types of immune cells have vitamin D receptors. At sufficient levels, the vitamin promotes differentiation of monocytes to macrophages, and increases their ability to destroy pathogens. It increases antigen presentation, but tempers cytokine release, possibly preventing cytokine storms.
Leo Galland, MD, a functional medicine practitioner in New York City, points out that vitamin D is essential for normal function of the Angiotensin Converting Enzyme-2 (ACE-2), and that deficiency impairs ACE-2. This has implications for COVID, in that the virus binds to ACE-2 on the surface of alveolar endothelial cells. An overwhelming of normal ACE-2 activity is one of the hallmarks of the COVID syndrome. Vitamin D supplementation might, potentially, attenuate this.
Vitamin D metabolites prevent excessive expression of inflammatory cytokines while also stimulating expression of anti-microbial peptides from neutrophils, monocytes, natural killer cells, and lung epithelial cells (Cannell JJ, et al. Epidemiology & Infection. 2006).
On a population basis, there is a crude inverse correlation between vitamin D levels and susceptibility to COVID. In a very recent cross-sectional analysis, 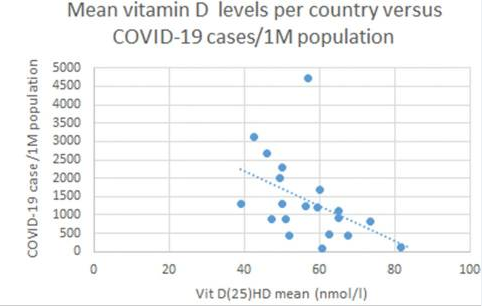 Petre Christian Ilie and colleagues showed that COVID prevalence and mortality were both highest in the countries with the lowest mean vitamin D. Portugal, Spain, Italy, and Switzerland were among the countries with the lowest mean vitamin D levels in COVID patients, and this was especially pronounced among the elderly.
Petre Christian Ilie and colleagues showed that COVID prevalence and mortality were both highest in the countries with the lowest mean vitamin D. Portugal, Spain, Italy, and Switzerland were among the countries with the lowest mean vitamin D levels in COVID patients, and this was especially pronounced among the elderly.
Ilie’s findings corroborate a 2013 systematic review by Jolliffe and colleagues showing consistent inverse associations between vitamin D status and risk of upper and lower respiratory infections.
Individual susceptibility may be partially explained by the fact that vitamin D deficiency impairs macrophage production of surface antigens, phosphatase, and H2O2, all of which play key roles in macrophage-mediated defense against pathogens (Abu-Amer Y, Bar-Shavit Z. Cellular Immunology. 1993)
Supplementation can reduce risk of acute respiratory infections (ARI), according to a massive metanalysis by Adrian Martineau of the London School of Medicine & Dentistry, who collaborated with researchers all over the globe to assess the preventive impact of vitamin D.
The Martineau paper, published in the British Medical Journal in 2017, covered 25 randomized vitamin D trials involving nearly 11,000 total patients. Irrespective of dose level or schedule, they found that vitamin D supplementation at any dose level translated into a 12% aggregate reduction in ARI incidence. There was a 19% reduction among people getting daily or weekly doses without bolus dosing.
Subjects with the lowest baseline 25(OH)-D levels benefit the most from supplementation. In people with baseline levels under 25 nmol/L, there was a 70% reduction in ARIs, compared with a 25% reduction among those with baselines above 25 nmol/L.
In reviewing the Martineau data, pulmonologist Roger Seheult notes that the number needed to treat (NNT) with vitamin D to prevent ARIs in a general 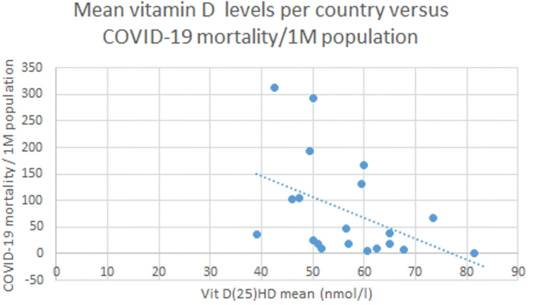 population is about 33, “roughly the same as for aspirin to prevent myocardial infarction.” Among people who are deficient, the NNT is 4, making supplementation a no-brainer.
population is about 33, “roughly the same as for aspirin to prevent myocardial infarction.” Among people who are deficient, the NNT is 4, making supplementation a no-brainer.
Seheult, a critical care specialist in Banning, CA, also pointed to The Irish Longitudinal Study on the Aging (TILDA), a prospective study of more than 8,000 Irish adults over age 50. The data suggest that at doses of 400 IU per day for low-risk individuals, and 800-1,000 IU per day for deficient and high-risk patients, vitamin D3 could reduce the prevalence of (non-COVID) chest infections by 50%.
Researchers at the University of Grenada recently launched a 10-week study to assess the impact of single 25,000 IU bolus dose of vitamin D in a cohort of 200 individuals with confirmed SARS-CoV-2 infections.
Calder and colleagues note that doses aimed at reducing respiratory infections are generally higher than the US RDA’s which range from 400–800 IU (depending on age). They recommend a daily intake of 2000 IU vitamin D3 per day to optimize immune function. IFM’s guidelines suggest going as high as 5,000 IU/day.
In a MedCram video detailing his personal supplementation protocol, Seheult—who treats COVID patients every day—says he sees little harm in upping the dose, especially for clinicians exposed to SARS-CoV-2. He’s taking 2,500 IU daily.
It is worth noting that the TILDA data show a strong inter-relationship between vitamin D deficiency, age, and obesity—all three are known risk factors for COVID morbidity and mortality, and they may be inter-related. Obese people were more likely than non-obese to be deficient, and this difference was more pronounced in people over the age of 70.
VITAMIN A
The IFM’s guidelines recommend supplementation with vitamin A, owing to its regulatory role in cellular and humoral immune responses. It affects T-helper cell function, production of secretory IgA, and regulates cytokine production.
In a review paper on potential interventions for COVID, researchers at the Shengjing Hospital in Shenyang Liaoning, China, point out that viral infections like measles tend to be worse in vitamin A-deficient children. Retinoids appear to, “inhibit measles replication is upregulating elements of the innate immune response in uninfected bystander cells, making them refractory to productive infection.” (Zhang L, Liu Y. J Med Virol. 2020)
Vitamin A supplementation can reduce morbidity and mortality in a range of human infectious diseases including measles-related pneumonia and HIV/AIDS.
IFM’s COVID recommendations suggest supplementation with 10,000 – 25,000 IU per day, though there are no clinical studies to support that.
Dr. Galland is more cautious. In his Coronavirus Handbook, he reminds colleagues that vitamin A (retinoids) can cause liver toxicity at high doses. He recommends supplements only if a patient shows low blood levels.
B VITAMINS & NAD+
In their recent paper, Zhang and Liu point out that people with deficiencies in B vitamins have weaker overall immune system responses to a range of pathogens.
In vitro experiments with human plasma show that vitamin B2 (riboflavin) can reduce the titers of MERS-CoV—the coronavirus pathogen that killed roughly 35% of all people infected with it back in 2012 (Kiel SD et al. Transfusion. 2016)
In mouse experiments and in vitro studies, vitamin B3 (nicotinamide) enhances the destruction of Staphylococcus aureus via a myeloid-specific transcription factor (Kyme P, et al. J Clin Investigation. 2012).
There is not a lot of specific evidence to make formal recommendations for B vitamins to prevent or treat COVID. But given their central role in hundreds of metabolic reactions, it makes sense to consider them, especially for elderly and at-risk individuals.
Several recent studies suggest that intracellular levels of nicotinamide adenine dinucleotide (NAD+), a vital metabolic co-factor, are depleted by infectious stressors, particularly by viral infections, and that supplementation to restore NAD+ may be beneficial. This phenomenon has been seen with a number of human viruses.
ARDS is characterized by a massive oxidative/nitrosative stress following viral entry into endothelial cells. This oxidative burst ultimately leads to apoptosis and necrosis due to NAD+ and ATP depletion.
In a preprint issued in March, geneticist Shirin Kouhpayeh and colleagues contend that NAD+ depletion “addresses all questions” in the COVID-19 infection process. They suggest that many of the pulmonary features of COVID, as well as the accompanying fatigue mood disorders, are due to depletion of NAD+, ATP, and serotonin—all of which depend on levels of vitamin B3, or its precursors like L-tryptophan.
Supplementation strategies aimed at boosting NAD+ might ameliorate the symptom burden. One possible approach is supplementation with nicotinamide riboside (NR)—an NAD precursor that can increase cellular NAD+ levels without causing flushing or inhibiting sirtuins.
VITAMIN E
Vitamin E compounds play a role in maintaining strong cellular immunity, particularly T-cell function, and especially in elderly people. T-cell decline is one of the most common features of immunosenescence.
“Both animal and human studies suggest that intake above currently recommended levels may help restore T-cell function which becomes impaired with aging,” reported Tufts University researchers Simin Meydani and Dayong Wu. “This effect of vitamin E can be accomplished by directly impacting T cells as well as indirectly, by inhibiting production of prostaglandin E2, a T cell-suppressing lipid mediator known to increase with aging.” (Wu D, Meydani SN. Endocr. Metab. Immune Disord. Drug Targets 2014).
Though there are not yet any COVID studies of vitamin E, data from earlier studies of other respiratory infections suggest that it might be beneficial. A randomized controlled trial of 617 residents of 33 Boston area nursing homes, daily supplementation with 200 IU vitamin E per day produced small but significant reductions in incidence of upper respiratory tract infections compared with placebo treatment (Meydani SN, et al. JAMA. 2004).
In 2009, De la Fuente and colleagues showed that at that same dose of 200 IU per day, vitamin E improved natural killer cell activity, neutrophil chemotaxis and phagocytosis, and mitogen-induced lymphocyte proliferation in elderly men and women (De la Fuente M, et al. Free Radical Res. 2009).
ESSENTIAL FATTY ACIDS
Omega-3 fatty acids, especially eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), may have a role in quelling the inflammatory response following infection with pathogens like SARS-CoV-2.
At the site of inflammation, EPA and DHA are enzymatically converted to compounds known collectively as specialized pro-resolving mediators (SPMs). These are a family of naturally occurring lipid mediators that play a crucial role in switching off the inflammatory response.
They do not block the initial inflammation phase, which is a desirable and usually beneficial response to infection or sudden injury. Rather, the SPMs down-regulate the process to resolve the inflammation once it has served its purpose. They function as “resolution agonists” targeting the immune cells that mediate the inflammatory response. By binding to specific cellular receptors, particularly the types known “G-protein coupled receptors,” SPMs modify cell behavior to promote resolution.
Calder and colleagues note that omega-3 deficiencies are associated with poor resolution of inflammation. “This could be very important in the context of severe COVID-19 which manifests as uncontrolled inflammation, the so-called cytokine storm linked with acute respiratory distress syndrome (ARDS).”
SPMs formed from EPA and DHA appear to be protective, at least in animal models of ARDS.
A recent Cochrane review of 10 clinical trials involving more than 1,000 hospitalized ARDS subjects did show significant reductions in mortality, ventilator days, and ICU lengths of stay in patients given tube-feeding formulas containing supplemental EFAs versus those given standard formulas.
The trial designs, trial quality, and EFA levels were so variable across the 10 studies that the Cochrane authors dismissed the findings, stating that it is unclear whether use of omega-3s confers any benefits in this context (Dushianthan A, et al. Cochrane Systematic Review. 2019).
None the less, Dr. Calder, who was one of the Cochrane authors, recommends intake of 250 mg/day of EPA + DHA for mitigating COVID risk.
ZINC
The value of zinc supplementation in preventing or mitigating viral infections is a contentious subject. Zinc is important for maintenance and development of cells in both the innate and adaptive immune systems. Deficiency impairs formation, activation, and maturation of lymphocytes, disturbs cytokine signaling, and weakens innate host defense, according to researchers at Aachen University Hospital.
A 2010 metanalysis of 10 randomized clinical trials involving a total of nearly 49,500 children concluded that routine zinc supplementation at doses ranging from 20 – 140 mg per week, reduced the incidence of acute lower respiratory infections.
The IFM COVID Task Force contends that zinc favorably modulates innate and adaptive immune system responses, and attenuates viral attachment, and replication. The guidelines advise supplementation with oral zinc acetate, citrate, picolinate, or glycinate, at levels of 30–60 mg daily, in divided doses, or as zinc gluconate lozenges.
In January, however, Finnish researchers questioned the efficacy of zinc acetate lozenges. They studied 253 City of Helsinki employees who self-identified as susceptible to severe URIs. Of the total cohort, 88 (35%) developed URIs during the study period, and they were more or less evenly divided between those randomized to placebo, and those using zinc lozenges six times daily for five days, for a total zinc acetate dose of 78 mg/day.
There were no differences between the groups in duration of URI symptoms or recovery rates (Hemila H, et al. BMJ Open 2020).
In his Coronavirus Handbook, Dr. Galland is skeptical about routine zinc supplementation.
“Zinc has been advocated at doses of 30 to 75 milligrams per day for its alleged direct anti-vital effects and for its inhibition of certain enzymes involved in viral transport and replication. This advice ignores the physiology of zinc. Levels of zinc in plasma, even when they are low, are about 10 times greater than those needed for inhibition of viral enzymes. The concentration of zinc inside cells is over 200 times higher than needed.”
He added that, “there is no way that zinc supplementation will impact the level of free intracellular zinc. But high dose zinc supplementation will produce deficiency of copper, and copper is a natural inhibitor of Furin.” The latter is an enzyme produced by all human cells, that SARS-CoV-2 hijacks in order to bind to the ACE-2 receptors on endothelial cells. Galland added that zinc supplementation raises the risk of bowel overgrowth with Clostridum difficile.
He believes supplementation only makes sense if testing shows serum levels to be low.
N-ACETYLCYSTEINE (NAC)
NAC is a major promotor of glutathione production, and there’s some evidence that it can mitigate the severity of viral infections. IFM’s Task Force recommends 600-900 mg of oral NAC twice daily.
Italian researchers studied the impact of 600 mg NAC, twice daily for six months versus placebo in a cohort of 262 individuals, most of whom were over 65 years of age. They report significant reductions in frequency and severity of influenza episodes, as well as fewer bedridden days in the NAC group. Local and systemic symptoms were sharply reduced, and though the frequency of seroconversion was similar in the two groups, only 25% of the virus-infected NAC patients became symptomatic, versus 79% in the placebo group (DeFlora S et al. Eur Respir J. 1997).







