 |
|
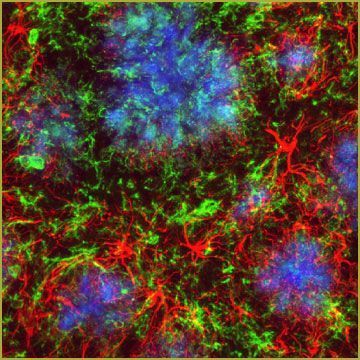
| A beautiful image of a horrible disease process: Neurotoxic β-amyloid plaques, stained blue, along with neurofibrillary tangles, are the hallmarks of Alzheimer’s disease. Neurologists know far more about the pathogenesis of this disease than in Dr. Alois Alzheimer’s day, but treatment options are still limited. New data suggest an extract of Angelica gigas, a plant used in Asian botanical medicine, may reduce production of β-amyloid and slow disease progression. Photo courtesy of Novartis. |
The patient, a 51-year-old woman, “showed jealousy toward her husband as the first noticeable sign of the disease. Soon a rapidly increasing loss of memory could be noticed. She could not find her way around her own apartment. She carried objects back and forth and hid them. At times, she would think that someone wanted to kill her and would begin shrieking loudly. She was totally disoriented to time and place. Occasionally, she stated that she could not understand and could not know her way around. Frequently, she shrieked with a dreadful voice for many hours. Her ability to remember was severely disturbed. The generalized dementia progressed, and after four and one half years of the disease, death occurred. At the end the patient was completely stuporous; she lay in her bed with her legs drawn up under her.”
It has been 100 years since Dr. Alois Alzheimer wrote the above description of a disorder that now bears his name. In the intervening century, neurologists have learned much about the pathogenesis of these symptoms. We’ve developed highly sophisticated imaging systems to display brain lesions in astonishing detail. Our care of people with dementia is, no doubt, more informed about the social and physical aspects than it was in Dr. Alzheimer’s day.
Unfortunately, though, our therapeutic options still fall far short of our patients’ needs. Too many people are dying in more or less the same way, with the same horrific symptoms, as Dr. Alzheimer described in 1907.
Up to 80% of all dementia fits Alzheimer’s pathologic description. Current estimates are that 10% of all people over age 65, and 40% of those over 85 suffer from Alzheimer’s disease (AD). There are roughly 4 million cases in the US, with 14 million expected by 2040. It is the 4th leading cause of death, and takes a fiscal toll of roughly $110 billion.
There haven’t been any shining advances in AD therapy in recent years. But new research focused on the inflammatory and oxidative aspects of the disease, and the emergence of an entirely novel botanical therapy derived from Angelica gigas (Korean Angelica) plant, are reasons to be hopeful.
Plaques & Tangles
Dr. Alzheimer accurately described the dying off of brain cells and consequent brain atrophy we now see directly with brain imaging. He made note of the round amyloid plaques, the twisted intracellular microtubules we now call neurofibrillary tangles, and the neuritic plaques composed of amyloid surrounded by dystrophic neuritic processes. In fact, it was only about 20 years ago that neurologists made any significant advances beyond what he’d described.
What we now know is that the plaques, mainly found in the hippocampus, amygdala, nucleus basalis of Meynert, locus ceruleus, dorsal raphe nucleus and neocortex, are composed of a specific protein called β-amyloid, the product of abnormal cleavage from amyloid precursor proteins. The insoluble fragment called Aβ1–42 promotes inflammation and oxidation, causing cell death and glial activation.
Neurofibrillary tangles are intracellular paired helical structures made of hyperphosphorylated tau protein. They correlate tightly with disease severity and neuronal death. Though we still don’t fully understand the role of β-amyloid and tau protein in causing the syndrome, there is evidence that both are neurotoxic.
Early AChievements
AD takes a particularly heavy toll on neurons using acetylcholine (ACh) as the signaling neurotransmitter. Lack of cholinergic activity in the brain’s neuronal networks is the overwhelming, though by no means isolated, neurochemical deficit in patients with AD. Specific brain nuclei with densely distributed cholinergic neurons are especially hard-hit. Given that cholinergic neural networks mediate memory and other cognitive functions, the effects are widespread and devastating.
Early therapeutic efforts were focused on enhancing ACh by supplying choline and lecithin, the biochemical precursors of ACh. This approach was modeled on the bold successes in treating Parkinson’s Disease with L-dopa, the precursor to the deficient neurotransmitter, dopamine. In a sense it prefigures the current interest in targeted amino acid therapy to treat neurobehavioral disorders (for more on this, join www.holisticprimarycare.net and search “neurotransmitter”).
Subsequent work suggested that a better way to increase ACh would be to inhibit acetylcholinesterase (AChE), the enzyme that degrades acetylcholine in the synapses. Clinical trials showed that patients treated with AChE inhibiting drugs consistently scored better on cognitive tests. This approach dominated AD treatment for decades, and most approved drugs for AD (Tacrine, Aricept, Exelon, Reminyl or Razadyne) are AChE blockers. Current treatment guidelines still call for these drugs, although the clinical efficacy is not very obvious in most cases.
Broadening Focus
Recently, the research and therapeutic focus in AD has broadened. Cell death in AD is thought to be related to a dysfunction of glutaminergic neurotransmission manifested as neuronal excitotoxicity. This new focus on the glutaminergic system, specifically the N-methyl d-aspartate (NMDA) receptors, is a major divergence from the cholinergic approach.
Memantine, a new drug that targets the glutaminergic NMDA receptors, is the first fruit of this line of thinking. It is able to inhibit the prolonged influx of calcium ions driving the neuronal excitotoxicity. Unfortunately though, most clinical trials have been negative, though a few studies show statistically significant differences between treated and untreated subjects. Memantine is sometimes combined with AChE inhibitors in the hope it will delay progression. The clinical efficacy of this drug, however, remains suspect.
A promising new direction is being forged by Korean researchers at the Hallyum University, Chuncheon and the Samsung Seoul Medical Centre, who are looking at a proprietary standardized extract from Angelica gigas, a plant commonly used in Asian botanical medicine. What is most interesting about this botanical is that it appears to reduce Aβ toxicity while also improving the cholinergic deficit.
Animal studies show that INM176, as the extract is known, can inhibit AChE, thus increasing ACh levels in the synapses, the desired effect of most of the AD drug therapies. More interesting is the data, again from rodent experiments, suggesting INM176 diminishes toxicity after intracerebroventricular injection of Aβ. Pretreatment with the Angelica extract improves the levels of ACh in mice injected with amyloid compared with control animals not receiving the extract. In mice with scopolamine or Aβ-induced amnesia, INM176 improved performance on passive avoidance tests and other behavioral measures.
The active components of INM176 have both antioxidant and anti-inflammatory properties, and they may actually reduce the production of Aβ from amyloid precursor proteins.
A recent clinical trial in Korea involved 80 elderly subjects with cognitive impairment randomized to 12 weeks of treatment with placebo or oral INM176. All subjects had a score of less than 25 on the Korean version of the Mini Mental Status Exam (K-MMSE) and were at high risk for AD based on scores on the 7-Minute Neurocognitive Screening Battery. The primary clinical measure was the AD Assessment Scale—Cognitive section (ADAS-cog).
After 12 weeks, the INM176 patients showed a marked decrease in ADAS-cog scores while the placebo group showed a slight increase. Mean changes in the Instrumental Activities of Daily Living (IDAL) and Geriatric Depression Scale (GDS) both favored the INM176 group.
While it is far too soon to draw definitive conclusions, the data definitely suggest that INM is effective in improving cognitive function, including memory, in patients on the fast track toward Alzheimer’s dementia.
INM 176 was developed for clinical use by Scigenic (www.scigenic.com) a Korean phytopharmaceutical company. It is available in the US as CognI-Q, from Quality of Life Laboratories (www.Q-O-L.com). It will likely be available in other branded professional-level nutraceutical products within the next 6 months.
So far, there have been no adverse effects from the Angelica extract, though admittedly, the trial was fairly small, and more research is clearly warranted.
Botanical approaches to treatment of AD are not new. In fact, galanthamine (Reminyl), an approved anticholinesterase drug for AD, was originally derived from daffodils, though the pharmaceutical form is synthetic. Ginkgo biloba has also shown small degrees of efficacy in helping reduce the symptoms of AD.
While the data on INM176/CognI-Q is still preliminary, it has several advantages over available remedies. It not only increases the synaptic acetylcholine, it also appears to alter the basic cause of neuronal cell death from Aβ. The ability to neutralize the toxicity of Aβ would clearly be a tremendous advantage over other treatments developed thus far.
Thomas Walshe, MD, is Chief of General Neurology at Brigham & Women’s Hospital, Boston, and Assistant Professor of Neurology, Harvard Medical School. He has conducted numerous clinical trials for the treatment of Alzheimer’s disease over the last 20 years.







